International Health Regulations - Joint External Evaluation of Canada Self-Assessment Report
April 20, 2018
Public Health Agency of Canada
Foreword
The world we live in today is characterised by the movement of information, products and people at unprecedented levels. While these exchanges have innumerable benefits, they also present new types of risks, particularly to health. It is imperative that risks be mitigated and managed by a strong health system with appropriate ties to the authorities responsible for emergency management, borders, and national security.
All 196 States Parties to the International Health Regulations (2005), including Canada, recognize that protecting health security requires all countries to establish public health core capacities to prevent, detect, and respond to significant events with health consequences. Over the last 20 years, infectious disease outbreaks including SARS, H1N1, Ebola in West Africa, and Zika, have amplified the need for a global approach to health security. Canada's own experience and participation in international response to public health emergencies has allowed us to see clearly the extent of the consequences public health emergencies can have on a society, and the pressing need to ensure measures are in place to prevent and mitigate these kinds of events. The Joint External Evaluation will provide Canada with an evidence-based set of recommendations to build national resilience and contribute to international health security.
Municipal, provincial, territorial, and federal levels of government have established innovative programs and capacities to protect the health of their residents and respond quickly to emerging risks. Completing an external review of Canadian core public health capacities is an opportunity to highlight our country's successes, but also to reflect on persisting challenges and imagine new ways of improving our systems.
We are proud to acknowledge the body of evidence gathered here through extensive collaboration with jurisdictional partners in health and other sectors. This report describes Canada's progress on 48 indicators across 19 technical areas. It provides a unique overview of Canada's health system and current state of public health preparedness. Our hope is that it will inspire future research and encourage innovation at local, provincial, regional and national levels.
Siddika Mithani, PhD
President, Public Health Agency of Canada
Dr. Theresa Tam
Chief Public Health Officer of Canada
Canada's health System
Canada is the second largest country in the world by total area covering 9.9 million square kilometres. Canada is home to 36.7 million people, more than 80,000 known wildlife species, and almost 200 million livestock. Although it has one of the world's lowest human population densities (4 people per km2), most Canadians (76%) live in urban centres along the border with the United States, with more than a third (36%) of the population living in the country's three largest cities: Toronto, Montreal or Vancouver.
Canada is a constitutional monarchy with multiple levels of government, including federal, provincial or territorial, and indigenous self-governments. Each level of government has its own areas of responsibility, but there are also areas where governments share responsibility. The federal government deals with areas of law listed in the Constitution Act, 1867 and that generally affect the whole country, such as national defence, foreign affairs, and Aboriginal lands and rights. Through "equalization payments" the federal government plays a role in addressing fiscal disparities between provinces and in ensuring that standards of health, education and welfare are the same for every Canadian.
Provincial and territorial governments have the power to make laws that affect their province or territory directly and to manage their own public lands. They are also responsible for health care delivery and education. Municipal governments run cities, towns or districts, including managing community water systems, local public land, and emergency first responders (police, fire protection, ambulances).
Indigenous self-government is part of Canada's evolving system of cooperative federalism and distinct orders of government. First Nations communities in Canada have a separate autonomous governance structure of elected band councils with responsibilities and authorities similar to those of municipal governments and with increasing powers over health in their communities.
Agriculture, immigration and health are some of the areas where the federal government and provinces and territories share responsibility.
Disease in Canada
In general, Canadians experience good health on a number of measures-almost 90% of Canadians report having good to excellent health, 92% say their lives are satisfying or very satisfying, and 70% of Canadians report having very good or excellent mental health. Canada's average life expectancy of 82 years ranks it among the healthiest nations in the world.
Canada has made great advances in preventing and controlling infectious diseases, through widespread improvements in hygiene and sanitation, water treatment systems, food safety measures, mass immunization programs, research into and development of new drugs, and education campaigns around safe sex, handwashing and safe food preparation. Canada also has better surveillance systems in place, providing a clearer picture of immunization rates and the distribution of diseases. Despite these advances, Canadians are still getting sick from infectious diseases and some of this sickness is long-term and treatment resistant, creating situations of vulnerability. In addition, some Canadians are not as healthy as others or are at a higher risk for poor health outcomes. Indigenous and low income households in Canada, for example, still live with higher rates of inadequate housing and food insecurity, compared to other Canadians. As well, Canada's geography, population distribution, and cultural differences create unique challenges to the delivery of health services in the country's northern, remote and rural communities.
While chronic diseases like diabetes, cancer, and cardiovascular disease are the leading causes of morbidity and mortality in Canada, infectious diseases continue to impose a significant burden on populations and health systems. Infectious disease priorities in Canada include, rates of antimicrobial resistance (AMR); vaccine coverage and the re-emergence of childhood vaccine-preventable diseases, such as measles and whooping cough; increasing rates of sexually transmitted and blood-borne infections; vector-borne diseases emerging as a result of climate change, such as Lyme disease; increasing rates of salmonella; and the disproportionately high rates of tuberculosis among foreign-born, First Nations and Inuit populations in Canada.
Although overall Canada's AMR rates are relatively low, there are upward trends in the rate of methicillin-resistant Staphylococcus aureus (MRSA) blood stream infection (BSI) in pediatric hospitals; in the rate of vancomycin-resistant Enterococcus (VRE) BSI in adult hospitals; and in the rates of drug-resistant gonorrhea in Canada.
The incidence of vaccine-preventable diseases in Canada is low. However, the viruses and bacteria that cause these diseases circulate within Canada and around the world and can still potentially cause outbreaks in under- and un-immunized groups. Since 2005 Canada has had a number of imported cases of measles resulting in spread and outbreaks within Canada. These importation events underline the ongoing risk of resurgence and the importance of achieving and maintaining Canada's vaccination coverage goals. Canada's public health and surveillance efforts have continued to maintain elimination of endemic measles in Canada. Raising immunization rates for measles, diphtheria, pertussis and tetanus in Canada-which currently are below national coverage goals of 97% by age two years-will further guard against the spread of these diseases following importation by travellers returning from endemic countries.
Government investments have contributed to the prevention and control of some sexually transmitted and blood borne infections. However, new HIV and hepatitis C infections continue to occur among certain populations and reported rates of chlamydia, gonorrhea and syphilis in Canada have been steadily rising since the late 1990s.
Climate change has been implicated in rising rates of vector‐borne communicable diseases in Canada. West Nile Virus appeared in Canada for the first time in 2002 and the incidence of Lyme disease has been increasing as higher temperatures allow mosquitoes and ticks to spread within Canada.
Serious outbreaks of food-borne diseases are rare in Canada. However, food-borne bacteria, parasites and viruses still cause illnesses in Canada. Every year, about 4 million (1 in 8) Canadians are affected by a food-borne illness. Over the last three years salmonella rates in Canada have increased mainly attributed to a rise in the incidence of salmonella enteriditis.
Canada has one of the lowest incidence rates of tuberculosis in the world at 4.4 cases per 100,000 population. Although the overall incidence of tuberculosis in Canada has steadily decreased over the last 30 years, tuberculosis continues to disproportionately affect First Nations and Inuit populations, with incidence rates of 20.4 per 100,000 and 198.3 per 100,000, respectively.
Diseases and conditions linked to unhealthy living (diabetes, obesity and mood disorders) have been increasing in Canada. Over a relatively short period of time, for example, the proportion of Canadians living with diabetes almost doubled from 6% in 2000 to 10% in 2011. This is a concern because type 2 diabetes is linked to higher proportions of people with an unhealthy diet, low physical activity and higher rates of obesity, all of which are linked to a variety of other diseases and conditions, making them proxies for overall health. Moreover, the gap between the highest and lowest income groups is widening. In 2014 Canadians in the lowest income group were twice as likely to report living with cardiovascular disease as those in the highest income group.
As a major exporter of live animals and animal products, and also a significant importer of animal products, and some live animals, Canada adopts a very rigorous approach to identifying and mitigating possible risks and has strict border controls in place. Despite the low levels of accepted risk, Canada has faced a number of major disease challenges in recent years including bovine spongiform encephalopathy, avian influenza and bovine tuberculosis. These outbreaks of foreign animal diseases have been effectively managed and the diseases either eliminated or are in the process of being controlled. Canada has also implemented a number of effective disease control and eradication programmes, including programs against tuberculosis and brucellosis.
Key features of Canada's health care system
Canada's core publicly funded health care system, known as Medicare, provides universal coverage for medically necessary hospital and physician services-patients do not pay user fees. While there are government programs that provide access to non-Medicare services for certain groups, such as children, seniors, and people with low incomes, the majority of working Canadians have private insurance plans to pay for services not covered by Medicare, such as prescription drugs outside of hospitals, vision care, dental care and physiotherapy. Quebec is the only province with a universal prescription drug plan. If neither public nor private insurance covers the full cost of a service, patients must pay out-of-pocket. While the Canadian health care system is mostly publicly funded through taxes, health care services are provided by a mix of public and private organizations and self-employed professionals.
The Canada Health Act sets out the criteria and conditions that provincial and territorial health care insurance plans must meet in order to receive the full federal cash transfer (Canada Health Transfer) to which they are entitled under the act. The five principles enshrined in the act are:
- Public administration: public health care insurance plans must be administered on a non-profit basis by a public authority
- Comprehensive: plans must cover all medically necessary hospital and physician services
- Universal: all eligible residents must be entitled to coverage on uniform terms and conditions.
- Portable: Coverage must be maintained when the insured person travels within Canada or abroad, within prescribed limits.
- Accessible: Reasonable access to insured health services must be maintained and unimpeded by financial or other barriers.
The act also discourages extra-billing and user charges for insured health services through mandatory dollar-for-dollar deductions from federal transfers.
Role of governments
Health care in Canada is primarily a provincial and territorial responsibility. Provinces and territories manage and deliver health care services in their jurisdictions (accounting for about 65% of Canada's total health expenditures). The federal government, through the Canada Health Act and other legislation, also plays an important role in matters that affect the health of Canadians, such as funding, regulating food and drugs, and setting national standards for health care.
Provinces fund and administer health insurance plans and other health care programs and they determine the organization and governance of their own health care systems. They regulate health care facilities and professionals, as well as private insurance; they manage capital investments; and they negotiate purchasing and pricing for their drug plans.
In provinces, regional health authorities plan, fund and deliver (within a defined geographical area) health care services, such as hospital care, rehabilitation and home care. However, regional health authorities are not responsible for physician services and drug plans, which remain the responsibility of provincial and territorial governments.
The planning and delivery of public health services in Canada is mostly done at the local or regional level through health departments of regional health authorities or districts or through health units and municipal health departments. These organizations have their own governance structures and their activities are governed by a provincial or territorial public health act (or equivalent) and its regulations, as well as by specific provincial or territorial legislation, policy, directives and conditions of funding--all of which vary from province to province. There is also considerable variation among public health units, which can serve populations from 600 to 2.4 million people with catchment areas from four to 800,000 square kilometers.
In addition, each province and territory has a chief medical officer of health (or equivalent) whose reporting relationship also varies considerably across the country as each province or territory tries to balance the independence of the CMOH as a health advocate with the need to integrate the portfolio into ministries of Health.
The federal government sets and administers national standards for Canada's health care system and funds provincial and territorial health care services through the Canada Health Transfer, an annual cash transfer to provinces and territories amounting to $37 billion in 2017-18 (about 23% of the total provincial and territorial health care expenditure). The federal government also regulates products, such as food, drugs, and pesticides, as well as medical and radiation-emitting devices; and it delivers or funds health care services to specific groups, including First Nations living on reserve, members of the Canadian Armed Forces, veterans, refugee claimants, and federal inmates. Indigenous Services Canada has a mandate to provide certain public health services to First Nations communities on reserve. Many Indigenous self-government agreements include the responsibility to deliver health care and public health services to their population.
All levels of government share responsibility for health care funding, health research and health promotion and protection, including emergency preparedness and response activities.
Funding
Canada spent about $242 billion on health in 2017. Although the system is predominantly publicly financed (70%), private financing (30%) plays an important role: private health insurance for services not covered by Medicare accounts for about 12% of Canada's health spending, while out-of-pocket payments by individuals for health services accounts for another 15%. Donations and other non-patient revenue streams make up the remaining 3% of private financing.
Besides the Canada Health Transfer to the provinces and territories, the federal government spent about $8 billion in 2017 (3% of total health spending) on direct health expenditures. Revenues for the publicly funded portion of health care expenditures come from federal, provincial and territorial tax revenues.
Health and medical research
Health research advances our understanding of the factors that influence health and plays an important role not only in improving health outcomes for Canadians but also in contributing to Canada's overall social and economic prosperity. Health research in Canada is supported by the federal and provincial governments, non-government organizations and industry. Most health research in Canada is conducted by the higher education sector (in association with research hospitals), industry and non-governmental organizations and some is conducted in the federal government's own facilities.
The Canadian Institutes of Health Research (CIHR) is Canada's major federal funder of health research. CIHR invests approximately $1 billion each year to support both investigator-driven (72%) and priority-driven (28%) health research in all four pillars: biomedical, clinical, health systems services, and population health. Chosen through a peer review process that ensures quality and fairness, top investigator-driven research proposals are funded through a variety of programs. Priority-driven research initiatives are created by the Government of Canada to investigate pressing health issues that are of strategic importance to the country.
CIHR's 13 institutes align their individual strategic plans with the overarching direction and goals of Canada's Health Research Roadmap II. Institutes work with stakeholders across disciplines, professions, sectors and geographic borders to identify health and health system needs and capture emerging national and international scientific opportunities.
Other federal funders of health research in Canada include the Natural Sciences and Engineering Research Council, Health Canada, Canadian Institute for Health Information, Genome Canada and the Canadian Foundation for Innovation.
Provincial research funding agencies also contribute to medical research and training in Canada through organizations such as Alberta Innovates, Fonds de recherche santé du Québec, Manitoba Health Research Council, Newfoundland and Labrador Centre for Applied Health Research, and British Columbia's Michael Smith Foundation for Health Research.
Self-assessment process
Canada announced at the North American Leaders' Summit in June 2016 its commitment to undergo a Joint External Evaluation (JEE) in June 2018. Project planning got underway in the fall of 2016, about 18 months before the planned site visit of the external evaluation team, and was achieved in four phases:
- Planning
- Stakeholder engagement
- Self-assessment
- External evaluation (site visits)
Led by the Public Health Agency of Canada (PHAC), the project involved 10 federal government departments and representatives from 13 provinces and territories.
A small project team was established within PHAC's Centre for Emergency Preparedness and Response. The project team worked with technical experts, policy analysts and senior managers within the Agency to design and hold stakeholder consultations, coordinate the gathering and validation of evidence for all 48 indicators, draft and produce Canada's self-assessment report, and plan and coordinate the external evaluation team site visit. (Table 1: Project Plan for Canada's Joint External Evaluation, June 2016 to June 2018)
The team identified technical leads-experts in each of the various technical areas-from PHAC, Health Canada, and the Canadian Food Inspection Agency to act as liaisons between the project team and program areas, to help gather data for the indicators, and to draft the self-assessment report's 19 technical chapters. Technical leads consulted with other experts within relevant federal program areas and with provincial and territorial colleagues, including a two-day meeting in November 2017 to discuss the evidence and to propose scores for each indicator.
| Phase | Activities |
|---|---|
| 01 Planning November 2016 to March 2017 |
Project team:
|
| 02 Stakeholder engagement January 2017 to March 2017 |
Project team:
|
| 03 Self-Assessment March 2017to December 2017 |
Project team:
Technical leads:
|
| 04 External Evaluation January 2018 to June 2018 |
Project team:
|
Self-assessment results
Indicator scores
Canada's Self-Assessment Report is an aggregate national assessment that was developed in consultation with federal, provincial and territorial governments. The indicator scores presented in table 2 below are the outcome of a two-day consultation with federal, provincial and territorial officials. The evidence in the technical chapters of this report and the proposed scores (on a scale of 1 "no capacity" to 5 "sustainable capacity") have been reviewed, validated and agreed upon by a wide range of government partners and technical experts.
| N/A | Indicator number | Description | Score |
|---|---|---|---|
| P1 Legislation | P1.1 | Legislation, laws, regulations, administrative requirements, policies or other government instruments are sufficient for implementation of IHR | 5 |
| P1.2 | The state can demonstrate that it has adjusted and aligned its domestic legislation, policies and administrative arrangements to enable compliance with the IHR | 4 | |
| P2 IHR Collaboration | P2.1 | A functional mechanism is established for coordination and integration of relevant sectors in the implementation of the IHR | 4 |
| P3 Antimicrobial resistance | P3.1 | Antimicrobial resistance detection | 3 |
| P3.2 | Surveillance of infections caused by antimicrobial resistant pathogens | 3 | |
| P3.3 | Healthcare associated infection prevention and control programs | 4 | |
| P3.4 | Antimicrobial stewardship activities | 3 | |
| P4 Zoonoses | P4.1 | Surveillance systems in place for priority zoonotic diseases/pathogens | 4 |
| P4.2 | Veterinary or animal health workforce | 4 | |
| P4.3 | Mechanisms for responding to infectious zoonoses and potential zoonoses are established and functional | 4 | |
| P5 Food safety | P5.1 | Mechanisms are established and functioning for detecting and responding to food borne disease and food contamination | 5 |
| P6 Biosafety and biosecurity | P6.1 | Whole-of-government biosafety and biosecurity system is in place for human, animal, and agriculture facilities | 5 |
| P6.2 | Biosafety and biosecurity training and practices | 4 | |
| P7 Immunization | P7.1 | Vaccine coverage (measles) as part of national program | 3 |
| P7.2 | National vaccine access and delivery | 5 | |
| D1 National laboratory system | D1.1 | Laboratory testing for detection of priority diseases | 5 |
| D1.2 | Specimen referral and transport system | 5 | |
| D1.3 | Effective modern point of care and laboratory based diagnostics | 5 | |
| D1.4 | Laboratory quality system | 4 | |
| D2 Real time surveillance | D2.1 | Indicator and event based surveillance systems | 5 |
| D2.2 | Interoperable, interconnected, electronic real-time reporting system | 4 | |
| D2.3 | Analysis of surveillance data | 5 | |
| D2.4 | Syndromic surveillance systems | 5 | |
| D3 Reporting | D3.1 | System for efficient reporting to WHO, FAO and OIE | 5 |
| D3.2 | Reporting network and protocols in country | 5 | |
| D4 Workforce development | D4.1 | Human resources are available to. Implement IHR core capacity requirements | 5 |
| D4.2 | Applied epidemiology training program in place | 5 | |
| D4.3 | Workforce strategy | 4 | |
| R1 Preparedness | R1.1 | Multi-hazard national public health emergency preparedness and response plan is developed and implemented | 5 |
| R1.2 | Priority public health risks and resources are mapped and utilized | 3 | |
| R2 Emergency response operations | R2.1 | Capacity to activate emergency response operations | 5 |
| R2.2 | Emergency operations centre operating procedures and plans | 5 | |
| R2.3 | Emergency operations program | 4 | |
| R2.4 | Case management procedures are implemented for IHR relevant hazards | 5 | |
| R3 Linking public health and security authorities | R3.1 | Public health and security authorities (law enforcement, border control, customs) are linked during a suspect or confirmed biological event | 4 |
| R4 Medical countermeasures | R4.1 | System is in place for sending and receiving medical countermeasures during a public health emergency | 5 |
| R4.2 | System is in place for sending and receiving health personnel during a public health emergency | 5 | |
| R5 Risk communications | R5.1 | Risk communication systems | 4 |
| R5.2 | Internal and Partner Communication and Coordination | 5 | |
| R5.3 | Public Communication | 4 | |
| R5.4 | Communication engagement with affected communities | 4 | |
| R5.5 | Dynamic listening and rumour management | 3 | |
| Points of entry | POE 1 | Routine capacities are established at points of entry | 5 |
| POE 2 | Effective public health response at points of entry | 5 | |
| Radiation emergencies | RE 1 | Mechanisms are established and functioning for detecting and responding to radiological and nuclear emergencies | 4 |
| RE 2 | Enabling environment is in place for management of Radiation Emergencies | 5 | |
| Chemical events | CE 1 | Mechanisms are established and functioning for detecting and responding to chemical events or emergencies | 4 |
| CE 2 | Enabling environment is in place for management of chemical events | 4 |
P1: National legislation, policy and finance
Joint external evaluation target: States Parties should have an adequate legal framework to support and enable the implementation of all of their obligations and rights to comply with and implement the IHR (2005). In some States Parties, implementation of the IHR (2005) may require new or modified legislation. Even where new or revised legislation may not be specifically required under the State Party's legal system, States may still choose to revise some legislation, regulations or other instruments in order to facilitate their implementation and maintenance in a more efficient, effective or beneficial manner. States Parties should ensure provision of adequate funding for IHR implementation through the national budget or another mechanism.
Level of capability in Canada
Canada implements the International Health Regulations (IHR) under existing legislation, regulations, policies and agreements in place at both the federal, and the provincial and territorial levels. An internal review conducted in 2010 found that the legislative and non-legislative measures taken by federal, provincial and territorial governments were sufficient to support implementation of the IHR in Canada.
As a federated state, Canada requires federal, provincial and territorial cooperation to implement the IHR. Provinces and territories have primary responsibility for health, public health and emergency response in their jurisdictions. Accordingly, they have their own legislation and regulations for governing these activities.
The federal government shares responsibility for public health with provinces and territories and delivers health care to specific populations (First Nations on reserve, Inuit, federal inmates and Canadian Armed Forces personnel). It also has responsibility and authority in sectors that affect public health. Examples of the core federal public health legislation and regulations are:
- Department of Health Act
- Public Health Agency of Canada Act
- Quarantine Act
- Human Pathogens and Toxins Act and Human Pathogens and Toxins Regulations
Federal legislation also governs sectors that affect public health, such as nuclear safety, radiation protection, food safety, and animal health.
National (federal-provincial-territorial) collaborating mechanisms, agreements, policies and plans are in place that clarify roles, help align legal and policy frameworks across jurisdictions, and ensure effective cooperation in emergencies. In particular, the Pan-Canadian Public Health Network plays a unique role in public health in Canada. It provides a national governance structure to support evidence-based decision making, information sharing and dissemination, and coordination and collaboration across jurisdictions. The Network has led to many important national agreements and plans, such as the Multi-lateral Information Sharing Agreement, the Federal, Provincial and Territorial Public Health Response Plan for Biological Events, and the Canadian Pandemic Influenza Plan for the Health Sector.
As well, Canada has a memorandum of understanding on the provision of mutual aid in relation to health resources during an emergency affecting the health of the public. The Operational Framework for Mutual Aid Requests for Health Care Professionals (OFMAR) puts the principles of the MOU into practice. This mutual aid agreement serves as an important means to ensure that all jurisdictions have the resources they need to respond to a public health emergency, including those that may not have the capacity to respond to a complex emergency.
Following the SARS outbreak in 2003, Canada strengthened its federal public health capacity by creating the Public Health Agency of Canada (the Agency) in 2004. Part of Canada's Health Portfolio, the Agency supports the Minister of Health in exercising the powers, functions and duties related to public health, which can be found in legislation such as the Department of Health Act. They can also result from programs approved by Cabinet and funded by Parliament through appropriations submitted for approval by the President of Treasury Board.
The Agency is the IHR National Focal Point for Canada. It facilitates coordination and collaboration of IHR implementation with provinces and territories and across the federal government. As an Agency program, the National Focal Point receives stable funding through the federal government's annual budgeting process. Canadian government organizations at all levels implement the IHR as part of their core mandate.
Canada is also an active participant in cross-border plans, agreements and networks (Global Health Security Initiative, North American Plan for Animal and Pandemic Influenza) that further, enable or strengthen national and international IHR compliance.
Government programs that deliver on IHR requirements are funded through regular annual budgeting processes. The federal government publishes online annual departmental reports on plans and priorities which must show budget allocations to support programs. Governments in Canada also make special funds available quickly to support emergency response activities.
Indicators
P1.1 Legislation, laws, regulations, administrative requirements, policies or other government instruments in place are sufficient for implementation of IHR
Canadian laws and regulations governing public health surveillance and response
Canada has an extensive range of legislation, regulations, policies and other instruments in place for governing public health surveillance, preparedness and response, and to support its compliance with the IHR and its ongoing strengthening of implementation.
Public health in Canada is a responsibility shared among federal, provincial, territorial and local governments. Provinces and territories have their own legislation governing emergency response. They also have public health legislation that establishes authority for public health surveillance and requires local public health staff to report notifiable diseases to public health officials. Provinces and territories, because they are responsible for delivering health services, are important partners and contributors of surveillance information in Canada.
The federal mandate to carry out surveillance is derived from the powers and obligations conferred on the Government of Canada by a number of acts, including the Department of Health Act and the Public Health Agency of Canada Act. The Department of Health Act gives the Minister of Health a broad mandate to protect Canadians against health risks and the spread of disease.
The Minister's duties, functions and powers include investigation and research into public health, including the monitoring of diseases and, subject to the Statistics Act, "the collection, analysis, interpretation and publication and distribution of information relating to public health."
The Public Health Agency of Canada was established to assist federal efforts to identify and reduce public health risk factors and to support national readiness for public health threats, including responding to a public health emergency. The Public Health Agency of Canada Act outlines the measures that the Agency can take in public health, including health surveillance and public health emergency preparedness and response.
The Public Health Agency of Canada Act mandates the Agency, in collaboration with its partners, "to contribute to federal efforts to identify and reduce public health risk factors and to support national readiness for public health threats." The act recognizes that public health surveillance is one of the public health measures that the Government of Canada undertakes through various programs and activities carried out by the Agency.
The Agency is responsible for assisting the Minister of Health in exercising or performing her functions relative to public health. Other departments within the Health Portfolio are Health Canada, the Canadian Food Inspection Agency, the Canadian Institutes for Health Research and the Patented Medicine Prices Review Board.
The Department of Health Act and the Public Health Agency of Canada Act do not expressly deal with the collection of personal information. However, under section 4 of the Privacy Act, the Agency can collect personal information for the purpose of carrying out programs and activities to assist the Minister in exercising her powers, duties and functions relative to public health, if the collection relates to that program or activity. Provinces and territories comply with the IHR obligation to disclose information for the purposes of managing a public health risk, in accordance with relevant domestic laws.
Other key federal legislation and regulations that enable Canada to meet its IHR obligations include:
- Quarantine Act: authorizes the measures that can be taken at points of entry or departure to control the spread of diseases that pose a significant risk to public health. This includes measures taken with respect to conveyances and their cargo.
- Potable Water on Board Trains, Vessels, Aircraft and Buses Regulations: governs the safety of water for drinking, hand washing, and preparation of food on certain conveyances.
- Human Pathogens and Toxins Act and Human Pathogens and Toxins Regulations: establishes a safety and security regime to protect the health and safety of the public against risks from human pathogens and toxins. The act takes a risk-based approach-identifying three risk groups-and covers obligations and prohibitions, licensing, standards, guidelines and enforcement.
- Food and Drugs Act and Food and Drug Regulations: establishes standards for the safety and nutritional quality of all foods sold in Canada. The act covers food content, as well as its manufacture, preparation, preservation, packaging and storing. It also authorizes enforcement measures, from inspections to seizures and destruction.
- Emergency Management Act: authorizes the Minister of Public Safety to coordinate emergency management activities with government institutions and in cooperation with the provinces and territories. This includes monitoring potential, imminent and actual emergencies and advising other ministers. The act also directs all federal ministers to identify risks in their area of responsibility and prepare emergency response plans for those risks.
- Health of Animals Act and the Health of Animals Regulations: protect animals and animal health, providing for the control of diseases and toxic substances that may affect land and water animals or that may be transmitted by animals to people. This includes segregation and inspection of animals, the importation of animal by-products, quarantine of imported animals, eradication of diseases, regulation of veterinary biologics, as well as permits and licencing and animal identification.
- Canadian Environmental Protection Act: aims to protect the environment and human health from pollution and toxic substances. The act covers pollution prevention through, among others, regulation of vehicle emissions and timelines for the management of toxic substances.
- Nuclear Safety and Control Act: establishes the Canadian Nuclear Safety Commission and authorizes it to regulate the development and use of nuclear energy and the production, possession and use of nuclear substances to limit the associated risks to national security, the health and safety of people and the environment.
- Nuclear Terrorism Act: strengthens authority to respond to nuclear terrorism threats and allows Canada to fulfill international commitments relative to nuclear security.
- Radiation Emitting Devices Act and Radiation Emitting Devices Regulations: authorizes Health Canada to set and enforce standards for the sale and use of these devices, including inspections, testing, and the provision of safety guidance.
- Transportation of Dangerous Goods Act: promotes public safety in the transportation of dangerous goods and covers, among other things, safety and security requirements, emergency response plans, containment and shipping rules and standards, inspection and monitoring, and certification.
- Hazardous Products Act: requires suppliers of hazardous products to communicate those hazards through product labels and safety data sheets as a condition of sale and importation.
- Privacy Act: protects the privacy of individuals with respect to personal information held by government institutions and covers the collection, use, disclosure, retention and disposal of that information in the administration of programs.
- Immigration and Refugee Protection Act has provisions related to the protection of public health (s. 38) and longstanding program structures for implementing these provisions, through health admissibility screening in the immigration system.
Policies and other government instruments
Because of the differences in legislation between the federal and provincial and territorial levels, Canada has mechanisms, agreements and plans in place (for example, the Health Portfolio Emergency Response Plan, the Canadian Pandemic Influenza Preparedness Planning Guidance for the Health Sector, and the Strategic Emergency Management Plan) that enable national coordination, particularly during public health emergencies that require federal involvement.
The Pan-Canadian Public Health Network is a national body that strengthens and enhances Canada's public health capacity and enables federal, provincial and territorial governments to work together on the day-to-day business of public health and to anticipate, prepare for and respond to public health events and threats.
The Network includes the Public Health Network Council, the Council of Chief Medical Officers of Health and three steering committees: Healthy Peoples and Communities; Public Health Infrastructure; and Communicable and Infectious Diseases. The Network was designed to support public health within a federated system where each level of government has its own area of responsibility. The Network's guiding principles are:
- Respect for the authority of each government to manage operations within their own domain
- Embrace differences in how governments exercise their public health responsibilities, set priorities and manage infrastructure
- Recognize that there is no "one size fits all" approach to public health
- Collaborate with non-governmental organizations
The Network's newly developed Blueprint for a Federated System for Public Health Surveillance in Canada (Blueprint), is a framework and action plan to formalize key elements of public health surveillance in Canada, such as governance, standards, ethics, information sharing, and performance measurement.
Canada's Multi-Lateral Information Sharing Agreement came into force in 2014. It is a legal agreement that establishes standards and deals with the sharing, use, disclosure and protection of public health information for infectious disease surveillance and public health emergency response. Until all technical annexes to the Agreement are completed, parts of an earlier federal, provincial and territorial Memorandum of Understanding on the Sharing of Information during a Public Health Emergency are still in effect.
In addition, the IHR National Focal Point for Canada-a funded program within the Agency-has joined with other federal departments and agencies to put in place policy and administrative arrangements to implement the IHR. Among these is a memorandum of understanding between the Agency and the Department of National Defence allowing the Department of National Defence to inspect and issue ship sanitation control certificates and ship sanitation control exemption certificates for Canadian Armed Forces vessels.
The IHR National Focal Point did a Privacy Impact Assessment in 2016 to ensure its compliance with Canada's Privacy Act in its collection, retention and distribution of information as part of the requirements under the IHR. The Assessment did not identify any high-level risks.
The IHR National Focal Point is also coordinating the development of guidance on how international case and contact notices (that might be shared with other States Parties National Focal Points) are managed from receipt to retention and distribution, ensuring compliance with relevant Canadian laws and policies.
The Federal Nuclear Emergency Plan describes the Government of Canada's preparation and response arrangements for managing the radiological health consequences of a nuclear emergency. The Plan has provincial annexes for jurisdictions having nuclear power plants or ports visited by nuclear powered vessels. These annexes establish the link between federal and provincial nuclear emergency response organizations and capabilities.
Canada is a signatory to the Convention on Early Notification of a Nuclear Accident and has arrangements in place for timely notification of a nuclear accident to the International Atomic Energy Agency, and for coordinating with the IHR National Focal Point to report to the World Health Organization (WHO). (See also section RE: Radiation emergencies.)
Examples of cross-border agreements supporting health security
Canada is an active partner in several formal and informal cross-border agreements and networks that support the IHR, health security, and collaboration at the global, regional and sub-regional levels. These include the Global Health Security Initiative, the Global Health Security Agenda, and the North American Plan for Animal and Pandemic Influenza (NAPAPI).
NAPAPI supports IHR implementation. It is a framework for comprehensive health security across all relevant sectors in the North American region. Its function is to protect against, control, and provide a public health response to animal and pandemic influenza in North America while avoiding unnecessary interference with international travel and trade.
NAPAPI complements national emergency management plans in the United States, Canada and Mexico. It builds on the principles of the International Partnership on Avian and Pandemic Influenza and on the standards and guidelines of the World Organization for Animal Health, WHO, the World Trade Organization, and the North American Free Trade Agreement.
To further enhance regional cooperation for health security, the three countries also have a long-standing informal practice of supporting cross-border "collaboration and assistance" (IHR, article 44) by sharing notifications to WHO of potential Public Health Emergencies of International Concern (IHR, article 6) and routine technical reports for the purposes of public health follow-up.
P1.2 The State can demonstrate that it has adjusted and aligned its domestic legislation, policies and administrative arrangements to enable compliance with IHR
Evidence that IHR implementation has been effective in Canada
An internal capacity assessment in 2010-validated by legal counsel in 2015-found that Canadian legislation and regulations were sufficient for IHR compliance and therefore no changes were recommended for IHR implementation. However, Canada understands that the public health landscape is constantly evolving. Through routine performance evaluation processes, and occasional independent investigations, governments in Canada identify and address gaps and weaknesses in existing government instruments.
The Quarantine Act, updated in 2005, serves as a good example of this. Since its implementation, several public health events have underscored challenges regarding the act and increased expectations for federal preparedness and response. Through the Border and Travel Health Modernization Initiative - an opportunity to refresh Canada's approach, strengthen collaboration, and better support compliance with the IHR-Canada is reviewing its legislation and policy, as well as other instruments, and adapting its approach to reflect a changing reality.
Certain policy and administrative arrangements have also been made to improve compliance with the IHR. Following a large Salmonella outbreak in 2014, the Public Health Agency of Canada, the Canadian Food Inspection Agency and Health Canada developed a joint protocol for IHR communications related to food safety issues. The protocol describes how Health Portfolio partners share information with the International Food Safety Authorities Network and WHO during an event with international implications. The protocol also outlines roles and responsibilities and describes reporting mechanisms and requirements to address duplication of effort and to better align messages.
Similarly, protocols have been put in place with the IHR National Focal Point to align reporting requirements in the event that Health Canada's Radiation Protection Bureau reports a real or potential nuclear emergency to the International Atomic Energy Agency under the Convention on Early Notification of a Nuclear Accident.
Best practices, challenges, gaps and recommendations
The Public Health Agency of Canada was created in 2004-with input from provinces, territories, stakeholders, and Canadians-in response to growing concerns about the capacity of Canada's public health system to anticipate and respond to public health threats. The Agency and the Chief Public Health Officer of Canada provide a focal point for federal leadership in managing public health emergencies.
Several provinces (including British Colombia, Ontario, and Quebec) have created their own leadership mechanisms for public health events and emergencies. These centres of expertise for managing public health risks and threats have improved Canada's capacity and leadership in both domestic and international health security. This multi-level arrangement can be viewed as a best practice in a federated state, such as Canada, that relies on strong multi-sectoral engagement and collaboration.
The Agency uses a results-based management approach to support its public health interventions.
As part of this approach, the Agency incorporates specific indicators from the Joint External Evaluation tool and the IHR annual monitoring tool into its regular planning and reporting process. This approach promotes accountability and comparability for delivering essential public health functions. Further, it supports priority-setting and resource allocation, and reinforces the concept that IHR implementation is fully integrated into public health planning and reporting in Canada. The Agency and all other federal departments must report annually to the Canadian public on plans and priorities, expected results, expenditure plans, and performance measurement.
In Canada, implementation of the IHR is enabled through broad health and emergency management legislation. This is supplemented by policy and administrative instruments and accompanied by public health or health-related provincial and territorial legislation. No major legislative gaps have been identified that might prevent the full implementation of the IHR. Canada nonetheless recognizes that policy and administrative instruments may need to be developed or revised to:
- Enable better coordination between legal frameworks
- Strengthen or clarify roles and responsibilities
- Improve performance of activities supporting IHR implementation
- Help address action items resulting from this Joint External Evaluation
P2: International Health Regulations coordination, communication and advocacy
Joint external evaluation target: The effective implementation of the International Health Regulations 2005 (IHR) requires multi-sectoral/ multidisciplinary approaches through national partnerships for effective alert and response systems. Coordination of nationwide resources, including the sustainable functioning of a National IHR Focal Point, which is a national center for IHR communications, is a key requisite for IHR implementation. The National Focal Point should be accessible at all times to communicate with the World Health Organization (WHO) IHR Regional Contact Points and with all relevant sectors and other stakeholders in the country. States Parties should provide WHO with contact details of National Focal Points, continuously update and annually confirm them.
Level of capability in Canada
In July 2005, the Public Health Agency of Canada was designated as the IHR National Focal Point for Canada. It is comprised of an IHR Program team and an IHR Operations function and is supported by the Health Portfolio Operations Centre Watch Office (Watch Office). The Agency performs the four mandatory functions of the National Focal Point as required under the IHR and several additional activities outlined in the WHO National Focal Point Guide.
Canada regularly validates the capacity of its National Focal Point. This is done through informal internal monitoring and assessment, twice yearly communication tests conducted by the WHO Regional Contact Point, and the WHO annual self-assessment.
The success of IHR implementation in Canada relies on ongoing collaboration among relevant sectors on issues of shared responsibility. Several collaborative groups meet regularly to ensure Canada fulfills its IHR responsibilities. One example is the IHR Implementation Working Group, made up of technical and policy experts from the Health Portfolio and other relevant federal departments.
There is also a network of IHR Champions in relevant federal, provincial and territorial government departments. These ensure that the IHR are reflected in regular operational activities and policy development processes. When required, existing federal/provincial/territorial governance mechanisms, task groups and emergency response plans are leveraged to support IHR implementation.
Indicators
P.2.1 A functional mechanism is established for the coordination and integration of relevant sectors in the implementation of IHR
Structure of Canada's IHR National Focal Point
Canada's IHR National Focal Point coordinates the implementation of the IHR on behalf of the Government of Canada. IHR implementation is shared by federal, provincial and territorial governments. The IHR National Focal Point is an implementation hub, providing advice, policy recommendations, advocacy and training, and stakeholder outreach. It also coordinates IHR monitoring and evaluation activities, including annual reporting to the World Health Assembly on behalf of the Government of Canada.
The Watch Office assumes the operational function of the IHR National Focal Point through around-the-clock service as a communications hub. Specifically, the Watch Office coordinates urgent communications concerning the implementation of IHR articles 6 to 12. The team provides critical situational awareness by gathering, organizing and redistributing stakeholder information on public health threats and risks. They also maintain internal tools for IHR communication, such as protocols, distribution lists, and templates.

Figure 1: IHR National Focal Point organizational structure - Text description
Figure 1 figure depicts the organizational structure of Canada's IHR National Focal Point. At the top of the organizational chart is the Centre for Emergency Preparedness and Response. The Centre is comprised of three offices: the Office of Border Health and Travel Services, the Office of Emergency Response Services, and lastly the Office of Situational Awareness and Operations. Within the Office of Situational Awareness and Operations there are two key sections to note: Situational Awareness and the Health Portfolio Operations Centre. The IHR National Focal Point falls under the Situational Awareness section and The Watch Office falls under the Health Portfolio Operations Centre. The Office of Situational Awareness and Operations and the areas that stem from it are all coloured in orange to indicate that these areas work together to deliver IHR NFP functions and services. All other teams are displayed in the colour blue.
The Watch Office program achieves 24-hour coverage using two teams: day-time watch officers and after-hours duty officers. During an event or emergency, a dedicated event watch officer is assigned to monitor and triage all event-related operational communications. An IHR technical advisor, generally a public health or health professional, is also available to assist the National Focal Point with assessing and reporting events (using the Annex 2 decision instrument), and with other activities. Advisors are accessible both during and outside regular business hours. The NFP also regularly consult program staff with specific disease expertise. The Vice President of the Health Security Infrastructure Branch (Public Health Agency of Canada) provides oversight on IHR implementation as the IHR Responsible Person in Canada.
IHR National Focal Point staff work closely with stakeholders to align program, policy, operational, legal, and privacy considerations to ensure that Canada continues to meet its assessment and reporting obligations under the IHR.
IHR Champions are designated points of contact in relevant federal, provincial and territorial government departments. They are familiar with Canada's obligations under the IHR and promote and support IHR implementation within their jurisdictions. IHR Champions act as a conduit and contact point for regular information exchange between the IHR National Focal Point and other government stakeholders in Canada.
Collaboration among multiple sectors is facilitated by a working group composed of technical and policy experts from the Health Portfolio and other relevant federal departments. The group meets regularly to support initiatives related to the implementation of the IHR in Canada. It has contributed to a variety of implementation activities, including Canada's Joint External Evaluation, annual self-assessment reports to the World Health Assembly, and other international capacity building activities. The working group does not facilitate collaboration during health emergencies.
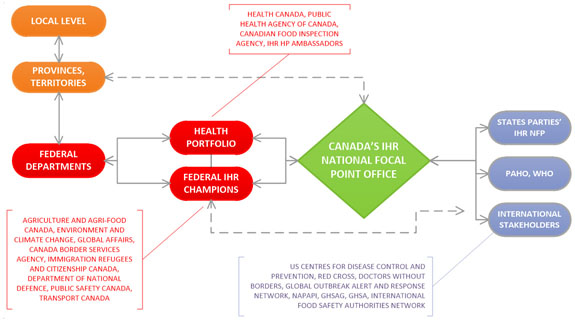
Figure 2: Coordination and information flow between Canada's National Focal Points and other sectors - Text description
Figure 2 diagram illustrates the flow of information and the coordination between Canada's National Focal Point and other sectors. In the top left corner in orange are two ovals representing jurisdictions across Canada. The first oval is the Local level and there is a two-headed arrow connecting it to the oval for Provinces/Territories below it, indicating a 2 way flow of information. The Provinces/Territories oval has a 2-headed dotted arrow connecting it to Canada's IHR National Focal Point Office in the centre of the diagram in a green diamond, and another 2-headed arrow connecting it to Federal Departments below it in a red oval, emphasizing the multi jurisdictional and multi-sectoral coordination that occurs.
To the right of the Federal Departments oval are two additional red ovals. The top one represents the Health Portfolio and the bottom one is Federal IHR Champions. The Health Portfolio is comprised of Health Canada, Public Health Agency of Canada, Canadian Food Inspection Agency and Health Portfolio IHR Ambassadors. Federal IHR Champions include representatives from: Agriculture and Agri-food Canada; Environment and Climate Change Canada; Global Affairs Canada; Canada Border Services Agency; Immigration, Refugees and Citizenship Canada; Department of National Defence; Public Safety Canada; and Transport Canada. There are 2-way arrows connecting the 3 Federal ovals, as well as an arrow leading from these federal entities to the IHR National Focal Point Office.
On the right side of the diagram are 3 blue ovals representing States Parties IHR NFP; PAHO / WHO; and International Stakeholders. International Stakeholders include US Centres for Disease Control and Prevention, Red Cross, Doctors Without Borders, Global Outbreak Alert and Response Network, NAPAPI, GHSAG, GHSA, International Food Safety Authorities Network. There are double sided arrows depicting information flow to/from these international bodies to Canada's IHR National Focal Point Office. There is also a double-sided dotted arrow leading back from these international groups to the Federal IHR Champions.
The Public Health Agency of Canada uses existing governance mechanisms to update partners in other relevant sectors on Canada's IHR-related activities. These mechanisms include the Pan-Canadian Public Health Network, the Council of Chief Medical Officers, the Public Health Infrastructure Steering Committee and the Communicable and Infectious Disease Steering Committee.
In addition to these day-to-day coordination mechanisms, Canada maintains several national emergency response plans (see section R1: Preparedness) and mutual aid agreements to improve response capacity (see sections R2: Emergency response operations and R4: Medical Countermeasures and personnel deployment).
Standard operating procedures, guidance documents and tools
As a best practice, Canada's IHR National Focal Point has developed a comprehensive standard operating procedure with two stand-alone components. One is a high-level strategic overview of Canada's IHR National Focal Point. It outlines its mandate and functions, stakeholder roles and responsibilities, and communication and coordination processes.
The other component is a detailed technical Guidebook for IHR Assessment and Reporting at the Federal (national) Level. This Guidebook supports the Health Portfolio program areas and technical experts in fulfilling their IHR duties to identify, assess, and notify WHO of certain public health events and other reporting requirements. It also includes useful information for all domestic stakeholders with IHR assessment and reporting responsibilities.
The National Focal Point also develops protocols, procedures, process flow maps, and templates to facilitate IHR communications and coordination. This includes the development of a guideline for sharing International Health Regulations notifications of events in Canada with the Council of Chief Medical Officers of Health and IHR Champions.
Individual Health Portfolio programs and other government departments are responsible for developing their own internal processes and procedures for detection, assessment, and reporting public health events, and for communicating relevant events to the IHR National Focal Point.
The IHR National Focal Point has developed additional communication tools to build an IHR community of practice in Canada. These include:
- Online educational resources for basic IHR understanding
- Training courses that provide in-depth operational knowledge and practice for IHR experts within the Health Portfolio
- Presentations to increase awareness of the IHR requirements and strengthen collaboration between sectors
Evaluation and testing of National Focal Point functions
Canada has confirmed its capacity to deliver the four mandatory core functions of a National Focal Point outlined in the IHR and WHO's guidance for National Focal Points in annual self-assessment reports to the World Health Assembly. In the 29th Pan American Sanitary Conference information document on IHR implementation, Canada is recognized as one of 12 States Parties in the Americas that have consistently submitted a State Party Annual Report since the requirement was instituted in 2011.
Canada's IHR National Focal Point performance is regularly monitored and assessed on an informal basis. For example, employees continually monitor the quality and timeliness of IHR communications. The team meets regularly to discuss operational issues, to review and revise internal IHR-specific processes, and to discuss ways to improve services.
Protocols are updated annually and as required. In addition, the National Focal Point staff host an on-going cycle of training and refresher sessions to raise awareness of IHR obligations and to ensure program standards are met. It also conducts regular outreach and provides training to other stakeholder groups (see D3: Reporting).
IHR coordination and communications are also reviewed following a response to an event or emergency. The results of these reviews inform the adjustment of internal protocols, procedures and practices to ensure they are suited to operational realities and emerging challenges.
To ensure the effectiveness of IHR communication functions, the Pan American Health Organization (PAHO), as the WHO IHR Regional Contact Point, conducts twice yearly tests with the designated IHR National Focal Point of States Parties in the Region. Canada's IHR National Focal Point has scored highly on all recent tests. No gaps or incidents of miscommunication have been identified. Nonetheless, these communication tests provide a valuable opportunity for the National Focal Point to review its internal processes and procedures, and to address any issues or deficiencies.
The Public Health Agency of Canada evaluated its emergency preparedness and response program to assess the program's core activities, including relevant IHR National Focal Point activities.
Best practices, challenges, gaps and recommendations
Over the past few years, Canada's IHR National Focal Point has focused efforts on building a foundation for its structure and processes within the Health Portfolio. As a result, Canada has a well-established and fully functional National Focal Point with the capacity to connect with WHO and partners for urgent IHR-related communications.
As a best practice, dedicated staff have been trained and made available to deliver on the National Focal Point's mandatory functions. Guidance documents and tools have also been developed to support these functions. One of the National Focal Point's key strengths is its streamlined approach to coordinating IHR-related communications and information flows.
IHR implementation is a shared responsibility in Canada. Consequently, the National Focal Point plans to strengthen partnerships between sectors by formalizing and standardizing collaboration and communication with federal, provincial and territorial stakeholders, including IHR Champions.
To do this, the National Focal Point will draw on the accumulated experience of the many sectors involved to improve IHR monitoring and evaluation activities, which include the annual reporting process. It will also keep stakeholders updated on implementation progress and establish clear links between health emergency management functions and IHR Champions in order to leverage parallel knowledge and expertise.
Ongoing outreach and training across government on IHR obligations and processes will therefore be key to ensuring that Canada continues to meet its requirements for IHR coordination, communications and advocacy. By building a strong domestic network or community of practice, the National Focal Point is laying the groundwork for open dialogue and information exchange among federal, provincial and territorial partners.
Canada's National Focal Point will also continue to work with PAHO/WHO, the United States, Mexico, and other partners on joint National Focal Point strengthening initiatives. Examples include peer-to-peer exchanges, training opportunities, and the development of valuable resources to help strengthen core capacities related to National Focal Point functions.
These opportunities foster a global community of practice and encourage open communications among National Focal Points. The result is more efficient and effective IHR-related communications within the Americas and beyond. This collaboration helps bolster overall health security and allows Canada to apply lessons learned from other countries to strengthening its National Focal Point capacities.
P3: Antimicrobial resistance
Joint external evaluation target: Support work being coordinated by the World Health Organization, the Food and Agriculture Organization of the United Nations, and the World Organization for Animal Health to develop an integrated global package of activities to combat antimicrobial resistance. The package spans human, animal, agricultural, food and environmental aspects (i.e. a One Health approach). These activities include:
- Each country having its own national comprehensive plan to combat antimicrobial resistance;
- Strengthening surveillance and laboratory capacity at the national and international level following agreed international standards developed in the framework of the Global Action plan while considering existing standards;
- Improving conservation of existing treatments and collaboration to support the sustainable development of new antibiotics, alternative treatments, preventive measures and rapid, point-of-care diagnostics, including systems to preserve new antibiotics.
Level of capability in Canada
In September 2017, the Government of Canada released a new framework, Tackling Antimicrobial Resistance and Antimicrobial Use: A Pan-Canadian Framework for Action. The Framework strengthens Canada's ability to combat the risks of antimicrobial resistance (AMR) in a coordinated and effective way
to minimize the impact of AMR, and to ensure that antimicrobials will continue to be an effective tool in protecting the health of Canadians. The Framework is grounded in a One Health approach, and developed with experts from the health, public health, veterinary and agriculture and agri-food sectors. Its four components are surveillance; infection prevention and control; stewardship; and research and innovation.
Addressing the threat of AMR in Canada requires the involvement of federal, provincial and territorial governments, health professionals, academia, industry, professional organizations (human and animal stakeholders) and the public. These groups must collaborate, coordinate and leverage activities across sectors to minimize duplication and to create effective, sustainable solutions.
AMR governance to guide the development of the Pan-Canadian Framework and Action Plan has three tiers: a Deputy Minister Champion Committee, an AMR Steering Committee, and four task groups (one for each of the Framework components). This structure has links to national health sector decision-making groups, such as the Public Health Network Council, the Council of Chief Medical Officers of Health, and the Conference of Deputy Ministers of Health. It also links to national agriculture sector committees through the Council of Chief Veterinary Officers, assistant deputy minister level regulatory and policy committees, and the federal, provincial and territorial ministers of agriculture. A federal interdepartmental committee (with representation from 11 departments and agencies) provides overall strategic direction and leadership for the Canadian response to AMR and for Canada's contribution to the international AMR agenda.
Canada has many surveillance systems in place to detect many of the AMR pathogens prioritized by the WHO. These systems include the Public Health Agency of Canada's nine national surveillance programs that track antimicrobial use (AMU) and AMR in both humans and animals. The data collected from these programs informs research and policy.
The Canadian Antimicrobial Resistance Surveillance System produces an annual report using information from national surveillance programs. Canada also has designated laboratories that can detect and report on AMR. One of these is the National Microbiology Laboratory, which is a Level 4 reference laboratory.
Each province and territory has accredited public health laboratories that can conduct AMR detection testing and submit isolates to the National Microbiology Laboratory for serotyping or susceptibility testing, and whole genome sequencing.
The Public Health Agency of Canada (PHAC) is the national coordinator for monitoring the incidence of AMR in bacterial isolates from human infections and in enteric bacteria along the food chain. The Canadian Nosocomial Infection Surveillance Program is responsible for clinical surveillance. The Program encompasses 65 acute-care hospitals which serve as sentinel surveillance sites for many healthcare associated infections, including infections due to Clostridium difficile and antibiotic-resistant organisms such as methicillin-resistant Staphylococcus aureus. Canada also collects some data on antibiotic-resistant organisms in community settings, including long-term care facilities.
For surveillance along the food chain, the Canadian Integrated Program for Antimicrobial Resistance Surveillance collects AMU data (national sales data, and farm-level data), and gathers cecal and other samples for AMR testing from farms, abattoirs, and retail food isolates. It also does AMR testing on clinical Salmonella isolates from animals and humans. Surveillance data from these systems is aggregated and publicly reported.
The delivery of health care in Canada is largely the responsibility of the provinces and territories. The federal government publishes evidence-based national guidance on infection prevention and control to inform and complement provincial and territorial guidelines, standards and protocols. This guidance is also used by different jurisdictions and facilities in the development of their policies and protocols.
In 2016 PHAC published Routine Practices and Additional Precautions for Preventing the Transmission of Infection in Healthcare Settings. PHAC is currently updating the 2002 version of the national guidelines on the prevention and control of occupational infections in health care. In addition to infection prevention and control policies, standards and plans for their health care facilities, most provinces and territories have access to designated infection control professionals on-site or through teaching hospitals.
The availability of isolation units in Canadian hospitals is high, although their capacity varies. Canada also has ad hoc measures in place to assess the effectiveness of infection prevention and control measures and share results.
As with health care, animal health care is largely the responsibility of the provinces and territories. Health Canada approves antimicrobials for sale for use in animals, whereas the provinces and territories control the distribution of antimicrobials and regulate the practice of veterinary medicine, At the farm level, National Biosecurity Standards and Biosecurity Principles, developed by the Canadian Food Inspection Agency (CFIA), in collaboration with producer organizations, provincial and territorial governments and academia, complement voluntary on-farm food safety programs.
The 2015 Federal Action Plan on Antimicrobial Resistance and Use in Canada: Building on the Federal Framework for Action includes commitments to strengthen the promotion of the appropriate
use of antimicrobials in human and veterinary medicine, and to continue to strengthen the regulatory framework for veterinary medicines and medicated feeds. Health care facilities across Canada have implemented antimicrobial stewardship programs and identified best practices. Veterinary Oversight of Antimicrobial Use – A Pan-Canadian Framework for Professional Standards for Veterinarians, guidelines and standards are in place to support the prudent use of antimicrobials in animals.
Health Canada monitors antibiotic sales and the number of prescriptions for human drugs using data collected from community pharmacies and hospitals, which it purchases from IMS Health Canada Inc. PHAC analyzes this antibiotic use data. Pilot studies are underway to improve our understanding of prescription practices for certain classes of antibiotics and select indications.
Health Canada regulates and approves veterinary drugs. Policies and regulations outlined by Health Canada related to feed are implemented and regulated by CFIA. PHAC analyses data provided by the Canadian Animal Health Institute on the annual volume of veterinary antibiotics distributed for sale in Canada. Additionally, Canada collects data on antimicrobial use at the farm level through the Canadian Integrated Program for Antimicrobial Resistance Surveillance sentinel farm program.
Health Canada maintains a Prescription Drug List, which is a list of medicinal ingredients that, when found in a drug, require a prescription for use in humans and animals. All systemic antibiotics in humans require a prescription in Canada, and all medically-important antibiotics for use in animals will require a prescription as of December 1, 2018.
Indicators
P.3.1 Antimicrobial resistance detection
National AMR plans
In September 2017, the Government of Canada released Tackling Antimicrobial Resistance and Antimicrobial Use: A Pan-Canadian Framework for Action. The Pan-Canadian Framework provides an overarching policy frame that lays out strategic goals and guiding principles to address antimicrobial resistance (AMR) in Canada. Specifically, it outlines the need for action across all jurisdictions and implicated sectors in the areas of: surveillance; stewardship; research and innovation; and infection prevention and control.
The Pan-Canadian Framework builds on previous plans that were developed to address AMR. In October 2014, the Government of Canada released Federal Action Plan on Antimicrobial Resistance and Use in Canada: Building on the Federal Framework for Action, which maps out a coordinated, collaborative federal approach to responding to the threat of AMR. In March 2015, the Federal Action Plan on Antimicrobial Resistance and Use in Canada: Building on the Federal Framework for Action was published. It identifies concrete steps to be taken by key federal departments and agencies to achieve the goals of the federal framework, including efforts to establish and strengthen surveillance systems.
Surveillance programs for AMR bacteria in Canada
Surveillance is a shared jurisdictional responsibility in Canada. There are data sharing agreements in place between federal, provincial and territorial partners. Surveillance is also one of the four pillars of the Pan-Canadian Framework. There are multiple surveillance systems in place at different levels of government in Canada that collect data on anti-microbial resistance and anti-microbial use (AMU) in human and animal settings, such as hospitals, community settings, agricultural settings and farms. The data from these systems is used to inform and update AMU and infection control policies.
- The Canadian Antimicrobial Resistance Surveillance System provides an integrated picture of AMR and AMU in Canada based on surveillance data from laboratory reference services and PHAC's surveillance systems that track identified priority organisms and AMU in humans and animals.
- The Canadian Integrated Program for Antimicrobial Resistance Surveillance monitors trends in AMU and AMR in selected bacterial organisms from human, animal and food sources across Canada. The program is based on several representative and methodologically unified surveillance components. These are linked to examine the relationship between antimicrobials used in food-animals and humans and the associated human health impacts. This information supports the creation of policies to control AMU in hospital, community and agricultural settings and the identification of appropriate measures to contain the emergence and spread of resistant bacteria between animals, food and people in Canada.
- In Canada, the list of nationally notifiable diseases for humans covers infectious diseases that have been identified by the federal government and all provinces and territories as priorities for monitoring and control efforts-this includes resistant microbes. Through the Canadian Notifiable Disease Surveillance System, provinces and territories voluntarily submit annual notifiable disease data which are used to produce national disease counts and rates.
The list of nationally notifiable diseases is revised periodically. Since 1987, all decisions about what diseases to include in the list are based on federal, provincial and territorial consensus, using set criteriaFootnote 1. Data from the Canadian Notifiable Disease Surveillance System is available to the public through Notifiable Diseases Online.
Laboratory capacity for AMR detection
The National Microbiology Laboratory is Canada's National Reference Laboratory and the designated laboratory for many of the Public Health Agency of Canada's AMR surveillance programs. The Laboratory is Canada's only Level 4 reference laboratory and is accredited by the World Organisation for Animal Health for Salmonella. The Laboratory validates its data using internal quality controls-control strains with a known minimal inhibitory concentration range and negative control. The Laboratory holds accreditation to ISO 15189 and ISO 17025 for some, but not all, tests. Many tests performed at the Laboratory are verified annually through participation in external proficiency tests or inter-laboratory comparisons.
Provinces and territories have public health laboratories, many of which can conduct AMR detection testing, or alternatively, submit isolates to the National Microbiology Laboratory for serotyping or susceptibility testing. The AMR pathogens for which laboratories can test vary from facility to facility due to surveillance priorities and testing methods within each province and territory.
The CFIA operates laboratories for pathogen testing in food, animals, and animal feed. It types every pathogenic isolate for the presence of AMR markers using whole genome sequencing techniques. All CFIA laboratories are ISO 17025 accredited and routinely participate in external proficiency tests on inter-laboratory verification.
Clinical laboratories in Canada are certified through a variety of accreditation programs and there are several external quality assurance bodies in Canada and in North America to ensure quality testing and reporting of data. Routine antimicrobial susceptibility testing methods are aligned with the Clinical and Laboratory Standards Institute and/or the European Committee on Antimicrobial Susceptibility Testing Guidelines. Automated testing methods or testing on new antimicrobials follow the U.S. Food and Drug Administration guidelines, if available. All hospital and provincial clinical laboratories are certified through the College of American Pathologists accreditation program.
AMR reporting activities
The annual Canadian Antimicrobial Resistance Surveillance System report integrates AMR and AMU data from human and food chain surveillance systems. This includes information on surveillance data, initiatives and gaps, and the list of priority resistant pathogens to monitor on a national level in humans and animals. Data obtained from the Canadian Antimicrobial Resistance Surveillance System, the Canadian Integrated Program for Antimicrobial Resistance Surveillance, and the Canadian Nosocomial Infection Surveillance Program are communicated to stakeholders on request, and also made available to the public online.
All certified clinical laboratories in Canada report data on individual patients to physicians for patient care, while some provincial and private animal health laboratories submit clinical and non-clinical Salmonella isolates to the National Microbiology Laboratory for serotyping. Some provinces collect specific AMR pathogen information from hospital laboratories and animal health laboratories. However, this varies from province to province.
P.3.2 Surveillance of infections caused by antimicrobial-resistant pathogens
Clinical surveillance programs
- The Canadian Antimicrobial Resistance Surveillance System collects and reports information on human and animal AMU from PHAC's surveillance systems, and from laboratory reference services. The System also collects information on antimicrobial resistant organisms in community settings, including long-term care facilities.
- The Canadian Nosocomial Infection Surveillance Program collects national data on various healthcare associated infections, including infections from antimicrobial resistant organisms. The Program is the only national surveillance system for identifying and monitoring rates and trends in national and regional hospital-associated infections. These include antimicrobial resistant organisms and their strain types and resistance patterns, some of which have been identified as priorities for AMR surveillance by the Pan-Canadian Public Health Network and Communicable and Infectious Disease Steering Committee's Antimicrobial Resistance Surveillance Task Group.
As of 2017, the Canadian Nosocomial Infection Surveillance Program conducts surveillance on:
- Hospital-associated Clostridium difficile infections
- Methicillin-resistant Staphylococcus aureus infections, including healthcare- and community-associated methicillin-resistant Staphylococcus aureus and methicillin-resistant Staphylococcus aureus bacteremia
- Vancomycin-resistant enterococci infections
- Carbapenemase-producing organisms, including carbapenemase-producing Enterobacteriaceae and carbapenemase-producing Acinetobacter
- Antimicrobial resistant organisms related to central line-associated bloodstream infections in intensive care units and hip and knee surgical site infections
- There are 551 acute-care hospitals in Canada, according to information from the Canadian Institute for Health Information. As of December 2016, the Canadian Nosocomial Infection Surveillance Program was conducting surveillance in 65 acute-care hospitals in 10 provinces in Canada. Of these, 13 are large, acute, tertiary care hospitals with more than 500 beds; 32 of the acute-care hospitals are intermediate size (201 to 500 beds) while the remaining 20 are smaller acute-care facilities with less than 200 beds.
- The only animal clinical pathogen that the Canadian Integrated Program for Antimicrobial Resistance Surveillance monitors is Salmonella, which is received from passive surveillance of veterinary diagnostic laboratories across Canada. National representation of human clinical Salmonella is obtained from provincial and territorial public health laboratories. Human clinical specimens of Campylobacter are collected for AMR testing through a partnership with a sentinel surveillance program called FoodNet Canada.
In addition to participating in the Canadian Nosocomial Infection Surveillance Program, many health care facilities have their own surveillance and monitoring programs in place. For example, Manitoba reports that all of the five regional health authorities and the mental health centre conduct surveillance for AMR organisms. Alberta, British Columbia, and Ontario report that all of their acute-care hospitals conduct surveillance for AMR pathogens in humans.
Quebec has a provincial surveillance program (Available in French only) for nosocomial infections, including several AMR pathogens. The participation of short-term hospitals in these monitoring programs is mandatory. The Yukon Hospital Corporation tracks rates of methicillin-resistant Staphylococcus aureus, vancomycin-resistant enterococci and Clostridium difficile across the territory's three hospitals. The Stanton Territorial Hospital serves as the sentinel site for the Northwest Territories.
Animal and food chain surveillance programs
- According to the Archived: 2016 Census of Agriculture, there are 77,594 livestock and poultry farms in Canada, as classified by the North American Industry Classification System. Of these, 57,802 are cattle, sheep, goat, pig, or poultry (chicken and turkey) farms. From this total, 309 are selected to represent their respective commodities, acting as Canadian Integrated Program for Antimicrobial Resistance Surveillance sentinel sites for the surveillance of AMR enteric bacteria in livestock on farms. As of May 2017, there were 90 sites for pigs, 129 for broiler chickens, 78 for turkeys, and 12 for beef cattle (large feedlots). Samples at these sites are collected to isolate foodborne enteric bacteria (E. coli, Salmonella, and Campylobacter).
- The Canadian Integrated Program for Antimicrobial Resistance Surveillance also includes national data from cecal samples obtained from beef, swine and chickens at federally registered slaughter facilities across Canada.
- The Canadian Animal Health Surveillance Network is a collaborative federal-provincial-academic network that links veterinary laboratories across the country to collect animal surveillance data. The network also serves as a surveillance system for animal disease threats to animal and human health and to the security of the food supply.
P.3.3 Healthcare associated infection prevention and control programs
Infection prevention and control in the context of AMR focuses on non-antimicrobial strategies that prevent infections in human healthcare, community, veterinary and agriculture settings. In settings where healthcare is delivered, infection prevention and control programming is essential. This applies particularly to hospitals due to the risk of acquiring hospital-associated infections such as:
- Methicillin-resistant Staphylococcus aureus
- Carbapenem-resistant Enterobacteriaceae
- Multidrug-resistant Enterobacteriaceae
- Multidrug-resistant Candida auris
- Vancomycin-resistant enterococci
Infection prevention and control is another pillar of the Pan-Canadian Framework.
Healthcare associated infections surveillance and monitoring
The federal government conducts healthcare associated infections surveillance through the Canadian Nosocomial Infection Surveillance Program, which had 65 active sites as of December 2016. The Program sites share surveillance information (reports, data) through the Program network and with provincial and territorial partners (Ministers of Health) and the federal government to identify emerging trends and outbreaks. The federal government publishes Program reports on its website. Researchers present Program data at conferences, such as the annual Association of Medical Microbiology and Infectious Disease [Canada] meeting, and they publish papers on the data in peer-reviewed journals.
Provinces and territories also monitor high-risk groups for healthcare associated infections. For example, New Brunswick reports that one regional health authority conducts screening for methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci in dialysis and extended care units every six months, which will soon be accelerated to every three months. One facility conducts extended-spectrum beta-lactamase screening in dialysis units every three months. The other regional health authority screens high-risk admissions for targeted antimicrobial resistant organisms, while in both health authorities, neonatal units participate in the Canadian Neonatal Network, reporting positive blood and cerebrospinal fluid cultures.
Healthcare-associated infection plans and programs
The federal government develops national evidence-based guidelines and makes them publicly available to healthcare settings. The guidelines inform and complement guidance, standards and protocols developed by provinces and territories, regional health authorities and healthcare facilities, and guidance documents developed by other organizations.
The federal government has published a series of Infection Control Guidelines to help healthcare organizations, infection control professionals and other healthcare providers develop policies and procedures related to infection prevention and control practices in healthcare settings. Within this series are guidelines on Nosocomial and Occupational Infections that address specific diseases, including Clostridium difficile in both acute-care and long-term care facilities, and Carbapenem-resistant Gram-negative bacilli. There is also guidance on Routine Practices and Additional Precautions for Preventing the Transmission of Infection in Healthcare Settings.
The federal government manages an expert advisory body for infection prevention and control, and engages with provinces, territories and stakeholders. Policy activities take place at the AMR and healthcare associated infections interface. For example, the Canadian Nosocomial Infection Surveillance Program works with the Canadian Hospital Epidemiology Committee of the Association of Medical Microbiology and Infectious Disease Canada on AMR pathogens that should have data collected at the hospital level.
Other groups develop standards informed by federal guidance:
- Infection Prevention and Control Canada and the Association of Medical Microbiology and Infectious Disease Canada are two groups that are formally recognized as "liaison member organizations" of PHAC's Infectious Diseases Prevention and Control external advisory body.
- Canadian Standards Association develops and maintains standards for infection prevention and control, sterilization, medical devices, and health care facility infrastructure.
- Accreditation Canada develops required operational practices for infection prevention and control.
- Canadian Patient Safety Institute has developed a number of tools for infection prevention and control, including a "Getting Started Kit", videos, and hand hygiene programming. The Institute collaborates with PHAC and other partners on the annual STOP! Clean Your Hands Day campaign.
Provinces and territories design and implement their own infection prevention and control strategies, which vary in scope and coverage. For example, in 2006, the Quebec ministry of health and social services (MSSS) established a reference framework for healthcare establishments for the prevention and control of nosocomial infections that defines procedures for combating nosocomial infections. The framework was updated in 2017.
In addition, every health care facility in Quebec must develop a nosocomial infection prevention program covering six components:
- Monitoring of nosocomial infections
- Policies, procedures and support measures
- Education and training
- Evaluation
- Communication and information
- Management of outbreaks
The content of the program must be adapted by each institution according to its size, mission, activities, and the priorities arising from local/regional epidemiology.
The federal government, provinces and territories have guidelines for protecting health care workers from occupational infections, including:
- Canada's national guideline on the Prevention and Control of Occupational Infections in Health Care. The guideline, which is being updated, supports collaboration between occupational health and infection control programs. It focuses on infectious diseases that are known to have been transmitted from person-to-person through occupational exposure, and on infections that may occur during the delivery of health care.
- Two Ontario guidance documents on the protection of health care workers from healthcare associated infections, which elaborate on the Ontario Occupational Health and Safety Act and regulations. Best Practices for Infection Prevention and Control Programs in Ontario explains the importance of adhering to the act and regulations and covers the use of personal protective equipment. Regulations pertaining to the proximity of food and drink to infectious materials; needle safety; and ceiling exposure values for biological and chemical agents. Routine Practices and Additional Precautions describes the requirements of the act and regulations and best practices for implementing them.
Trained infection control professionals
Dedicated infection prevention and control resources and programs vary across jurisdictions and centres. Teaching hospitals generally have higher proportions of infection control professionals and infectious disease specialists than community and long-term care settings. Infection prevention and control training is typically facility- or program-based.
British Columbia, Saskatchewan, Nova Scotia, and Newfoundland and Labrador report infection control professionals in all tertiary hospitals. Prince Edward Island has infection control professionals in all health authority hospitals, community hospitals, long-term care facilities, and community programs.
In Quebec, hospitals that provide tertiary care have infectious disease microbiologists assigned to infection prevention and control, some of whom have completed specialized training. Acute-care hospitals in New Brunswick fall within the mandate of the infection prevention and control programs in their regional health authority, including access to infection control professionals. Smaller and medium-sized hospitals, where infection control professionals may not be on site daily, have access to them via telephone or internet.
Hospital isolation units
The Canadian Standards Association Group has standards and codes for the design, construction and renovation of healthcare facilities. The national Infection Control Guideline series and the Routine Practices and Additional Precautions document include general recommendations for isolation units. These recommendations are implemented at the provincial and territorial level.
Most provinces have isolation units or rooms, and several have airborne infection isolation rooms.
For example, in Alberta airborne infection isolation rooms are available and are monitored for function. Contact and droplet protection is provided in isolation rooms (with anterooms), in private patient rooms, and in shared patient rooms using appropriate cohort precautions. Infection control professionals are available to all acute-care hospitals for consultation on management of patients that require isolation. Alberta Health Services has created an isolation reference guide for adults as part of their overall resource manuals for infection prevention and control.
New Brunswick has 111 stand-alone airborne infection isolation rooms located in hospitals of varying sizesFootnote 2. Saskatchewan and Newfoundland and Labrador report that their tertiary hospitals are equipped with isolation rooms.
Assessing the effectiveness of infection prevention and control practices
Many of the infection prevention and control programs at the provincial and territorial level include reporting requirements and/or evaluation components. For example, Quebec, as part of its Ministerial Action Plan 2015-2020 (Available in French only) on the Prevention and Control of Nosocomial Infections, is planning an evaluation of the clinical and economic impact of nosocomial infections and the effects of infection prevention and control measures on these infections in general and specialized care hospitals.
In addition, members of Institut national de santé publique du Québec (Available in French only) [Quebec national public health institute] committees-the Quebec Nosocomial Infections Committee (Available in French only) and the Provincial Committee for Surveillance of Nosocomial Infections (Available in French only) - publish scientific articles on the impact of the guidelines on the rate of nosocomial infections.
Ontario has a Provincial Infectious Diseases Advisory Committee that develops best practices, reports and recommendations on communicable diseases, immunization, infection prevention and control and surveillance. Prince Edward Island and Saskatchewan evaluate their infection prevention and control according to Accreditation Canada requirements.
P.3.4 Antimicrobial stewardship activities
National plans and programs
Antimicrobial stewardship is a focus area of the Pan-Canadian Framework. There are a myriad of other hospital, community and veterinary-based antimicrobial stewardship initiatives in Canada that promote appropriate AMU among those who prescribe and dispense antimicrobials and those who use them. These initiatives include:
- Regulatory frameworks in place to review and approve antimicrobials for human and animal use, most of which have prescription status in Canada. The main regulatory framework (the Food and Drug Regulations, under the authority of the Food and Drugs Act) consists of various regulatory provisions applicable to all drugs sold in Canada, including antibiotics.
- Health Canada monitors the safety of antimicrobials on the market and requests manufacturers to make changes to labelling as needed. The Archived: Protecting Canadians from Unsafe Drugs Act outlines measures to strengthen Health Canada's oversight on the safety of therapeutic products throughout their life cycle; to improve reporting of serious adverse drug reactions and medical device incidents; and to increase regulatory decision-making transparency.
- Many health care facilities nationwide implement antimicrobial stewardship as part of voluntary accreditation standards.
Canadian initiatives that support the prudent use of antimicrobials in humans include:
- A stakeholder-led action plan on antimicrobial stewardship: Putting the Pieces Together: A National Action Plan on Antimicrobial Stewardship.
- Antibiotic resistance information and awareness materials for Canadians and health care professionals.
- Pan-Canadian Public Health Network Communicable and Infectious Disease Steering Committee Task Group on Antimicrobial Use Stewardship.
- Community education program about handwashing and the responsible use of antibiotics, "Do Bugs Need Drugs?"
- Canadian Guidelines on Sexually Transmitted Infections is a resource for primary care and public health professionals for the prevention, diagnosis, treatment and management of sexually transmitted infections.
- Practical tools and information for practitioners, as well as webinars on antimicrobial resistance offered on Antibiotic Awareness Week Canada.
- Choosing Wisely Canada is a campaign to help clinicians and patients engage in conversations about unnecessary tests and treatments, and make smart and effective care choices
Prudent antimicrobial use in animals is supported through a variety of programs, such as:
- A new national framework, Veterinary Oversight of Antimicrobial Use: A Pan-Canadian Framework of Professional Standards for Veterinarians supports provincial and territorial veterinary regulatory bodies in developing regulations, guidelines or bylaws related to veterinarian oversight in the use of antimicrobials. The framework contributes to the development and implementation of uniform regulations across the country.
- The Canadian Veterinary Medical Association's Antimicrobial Prudent Use Guidelines for Beef Cattle, Dairy Cattle, Poultry and Swine. The guidelines are being updated to include small ruminants and companion animals.
- National on-farm biosecurity standards, protocols and strategies developed for all major livestock and poultry groups in collaboration with federal, provincial and territorial governments, producer organizations and academia. These standards are implemented on a voluntary basis by many producer associations and farmers.
- Voluntary on-farm food safety programs designed by federal, provincial and territorial governments, in consultation with food animal producers, are based on recognized standards and best practices and focus on food safety and animal care. These include prudent use of antimicrobials to promote good animal health. Examples include: the Canadian Cattlemen's Association's Verified Beef Production Plus Program; the Canadian Pork Council's Canadian Quality Assurance program; and the Dairy Farmers of Canada's Canadian Quality Milk program and "proAction initiative."
- Chicken Farmers of Canada's Antimicrobial Use Strategy.
- The health sections of the National Farm Animal Care Council Codes of Practice for the care and handling of farm animals are guidelines on appropriate infection prevention and control and treatment of disease.
- Recent changes to the Food and Drug Regulations to increase oversight of antimicrobials available for use in animals. The changes restrict who can import medically-important antimicrobials and prevent the importation of the active pharmaceutical ingredients in medically-important antimicrobials by producers for their direct use in food animals. They also restrict the personal importation of drugs for food-producing animals to those on List B and prohibit medically-important antimicrobial labels from including growth promotion claims.
- Canada has in place legislation and regulations pertaining to import, marketing authorisation, production, distribution, quality control, and prudent use of high-quality veterinary drug products, including antimicrobials. These rules support a system of inspection of the whole drug chain.
During the evaluation of drug submissions for new antimicrobials, there is a requirement for studies or data assessing the risks arising from the potential development and spread of resistance under the proposed conditions of the use of the product.
Antimicrobial drug product inserts and labels give precise information on conditions for use, linked to studies in the drug submission application (compliance with prescribed doses, treatment duration, withdrawal period, and specific warning statements in the event of antimicrobial resistance).
Antimicrobial use monitoring in human medicine
- Canada has initiated several pilot surveys of antimicrobial use, including the Fluoroquinolone Prescribing and Use in Canadian Primary Care Practice survey, which provides a better understanding how the fluoroquinolone class of antibiotics is prescribed in humans. Another initiative is a two-phase study by the Drug Safety and Effectiveness Network to evaluate usage patterns across Canada of fluoroquinolones in select indications where the benefit of using these drugs may be marginal.
- Health Canada monitors antibiotic sales and the number of prescriptions for human drugs, while the Public Health Agency of Canada analyzes antibiotic use data collected from community pharmacies and hospitals and purchased from IMS Health Canada Inc.
- Under the Food and Drugs Act, Health Canada maintains the Prescription Drug List. It is a list of medicinal ingredients that, when found in a drug, require a prescription. A prescription is required for the use of all systemic antibiotics in humans. Some topical antibiotics containing bacitracin, mupirocin, polymyxin B sulfate, or gramicidin do not require a prescription.
Antimicrobial use monitoring in food production, veterinary medicine and the environment
- Since 2006, the Canadian Integrated Program for Antimicrobial Resistance Surveillance has reported data from the Canadian Animal Health Institute on the annual volume of veterinary antimicrobials distributed for sale in Canada. Recent regulatory amendments require manufacturers and importers of veterinary drugs in dosage form that contain an active pharmaceutical ingredient for medically-important antimicrobials to report annually on the total quantity of each drug sold and provide an estimate of the quantity sold for each intended animal species. Individuals who import, compound and sell an active pharmaceutical ingredient for medically-important antimicrobials for veterinary use must also report on an annual basis.
- Most of the newer antimicrobials approved since 2004 for use in animals require a prescription. Canada is working to implement increased oversight by including the remaining medically-important antimicrobials for veterinary use on the Prescription Drug List. As of December 1, 2018, all such drugs for veterinary use will be sold by prescription only.
- The Canadian Integrated Program for Antimicrobial Resistance Surveillance monitors antimicrobial use at the farm level in sentinel sites from swine, broiler chickens, turkey, and is developing plans to monitor in other commodities.
Best practices, challenges, gaps and recommendations
Through the Pan-Canadian Framework for Action on AMR and AMU, Canada has established a foundation to identify steps to address key AMR and AMU challenges. The next phase will focus on creating a Pan-Canadian action plan, which will lay out concrete deliverables, timelines, measurable outcomes, priorities and allow for tracking progress.
The complexity of AMR underscores the need for coordinated action by key actors-governments, private and public partners, and the public-across the human, animal and environmental sectors. Canada has successfully brought together a variety of stakeholders engaged in fragmented activities across multiple sectors to work collaboratively. The goal moving forward is to maintain and enhance these activities.
Data collection from a range of healthcare, community, agriculture and veterinary settings is complex. Canada has developed a strong surveillance infrastructure for AMR. However, surveillance is a shared responsibility and there remain variations in how data are collected, managed and reported by multiple systems at the local, provincial, territorial and federal levels. In addition, resource and infrastructure capacity to collect and analyze data varies. Within this landscape, there is a need to ensure comprehensive data comparison and analysis of AMR and AMU.
Canada has done much to identify gaps related to its surveillance, stewardship, and infection prevention and control activities, as well as in the area of research and innovation (the fourth component of the Pan-Canadian Framework). Canada is committed to enhancing the strong One Health approach it has developed domestically to address AMR, and to leveraging it internationally.
Given the extensive consultations that have recently taken place in Canada during the development of the Pan-Canadian Framework, the recommendations outlined below focus on actions that will strengthen Canada's ability to reduce the risks of AMR in a coordinated and effective manner:
- Strengthen governance structures to generate knowledge and information on AMR and AMU in humans, agriculture and animals through the monitoring, detection and tracking of resistant organisms to develop and monitor interventions.
- Promote, facilitate and measure appropriate AMU in humans and animals to conserve the effectiveness of antimicrobials that are critical to human and animal health, and to limit the development and spread of resistant organisms within and among populations.
- Reduce the need for antimicrobial treatment by promoting infection prevention and control practices to decrease infection rates in healthcare, community and animal settings.
- Support the advancement of research and innovative approaches for the identification, characterization and real-time detection of microorganisms, including resistant bacteria, and the treatment and prevention of infections as well as basic and behavioural research.
P4: Zoonotic disease
Joint external evaluation target: Adopted measured behaviours, policies and/or practices that minimize the transmission of zoonotic diseases from animals into human populations.
Level of capability in Canada
In Canada, provincial and territorial governments and other animal and human health stakeholders routinely work with federal departments to prevent, detect and respond to outbreaks of zoonotic diseases of public health importance. Among these departments are the Public Health Agency of Canada (PHAC), the Canadian Food Inspection Agency (CFIA), Health Canada, and Environment and Climate Change Canada.
In Canada, enteric and non-enteric zoonoses are managed in separate but related streams. (For more information on enteric zoonoses see section P5: Food safety.) Canada is building its animal and human epidemiology, surveillance, response, and laboratory capacity at all levels. The focus is on non-enteric zoonotic diseases in both human and animal populations (including wildlife, pets, livestock and arthropod vectors).
The One Health approach, which is critical to the prevention and control of zoonoses, is generally accepted in Canada. Some national strategies take into consideration the human-animal-environment interface, which drives more and better collaboration across disciplines and sectors.
Domestic animal health is well managed and regulated at the provincial, territorial, and federal levels. Canada's wild animal populations are relatively large and dispersed. Consequently, responsibility for monitoring wildlife is a collaborative effort between several government and non-government organizations. These include provincial and territorial ministries of environment, conservation and natural resources; federal departments (such as Environment and Climate Change Canada, the Department of Fisheries and Oceans, Parks Canada); the Canadian Wildlife Health Cooperative; and Indigenous groups.
Canada has in place a number of well-established non-enteric zoonotic disease surveillance programs for disease occurrence in animal populations, including for West Nile virus, Lyme disease, rabies and animal (primarily avian) influenza. Provincial and territorial governments have primary responsibility to investigate illnesses and outbreaks of zoonotic diseases in humans, with support from relevant federal departments such as PHAC and the CFIA.
Government and non-government partners work together for a coordinated One Health approach. Canada's List of Nationally Notifiable Diseases covers zoonotic diseases in humans, while the CFIA's Federally Reportable, Immediately Notifiable and Annually Notifiable Diseases in Canada covers zoonotic diseases in animal populations.
At all levels of government there are strategies to guide emergency operations centres to coordinate an effective response to public health events, including zoonotic disease outbreaks. (See section R2: Emergency response operations.)
Canada has a sustainable, well-trained veterinary workforce of approximately 13,700 veterinarians. Five accredited colleges of veterinary medicine in Canada offer programs for pre-clinical and clinical study, as well as opportunities for advanced training and post-graduate study in public health and epidemiology. They have active research and teaching programs and operate veterinary diagnostic laboratories. Internship and fellowship programs are available in Canada to graduates of veterinary medicine and courses. These also extend to established professionals, for gaining new skills related to epidemiology, public health, and laboratory diagnosis for zoonoses in animals and people.
Indicators
P.4.1 Surveillance systems in place for priority zoonotic diseases and pathogens
Priorities for national zoonotic disease surveillance and control
The identification and prioritization of zoonotic diseases are brought forward via a number of mechanisms, including public health governance structures, programmatic analysis and decision-making and national technical informal working groups.
The Canadian Notifiable Disease Surveillance System is a federal, provincial and territorial collaboration that enables all levels of public health to share information on nationally notifiable infectious diseases. Provinces and territories use the System to submit annual notifiable disease data, which are used to produce national disease counts and rates. The Notifiable Disease Charts can be used to explore trends since 1924.
The List of Notifiable Diseases provides disease names and years in which they were considered notifiable. Public health departments use the information to monitor, prevent and control the occurrence and spread of all notifiable zoonoses, as well as other diseases and conditions. Non-enteric zoonoses under surveillance in Canada through the Canadian Notifiable Disease Surveillance System include: anthrax, brucellosis, hantavirus pulmonary syndrome, Lyme disease, malaria, plague, rabies, tularemia, viral hemorrhagic fevers, West Nile virus, and yellow fever.
The One Health concept is generally accepted in Canada and the integration of human, animal and environmental factors is regularly considered and applied in policy and program decisions. Pan-Canadian approaches reflected in strategies, such as the National Plant and Animal Health Strategy, the Pan-Canadian Framework on Clean Growth and Climate Change, and the National Wildlife Health Strategy (currently in development), all take into consideration the human-animal-environment interface. Canada also has several governance committees, councils and working groups listed under "Mechanisms in place to identify priorities" below. These bodies review and discuss issues related to One Health (see "Public health governance structures" below).
Several recent events in Canada have provided opportunities to test the skills of both human and animal public health workers in investigating and responding to a non-enteric zoonotic event:
- In September 2016 the CFIA began an investigation, still on-going, into cases of bovine tuberculosis in cattle on a farm in Alberta.
- In July 2016 authorities Archived: detected cases of H5N2 LPAI (low path) on a commercial duck farm in Ontario.
- In April 2015 there was an outbreak of H5N2 HPAI on a commercial poultry farm in Ontario.
- In December 2014 there was an outbreak of H5N2 HPAI on a number of farms in British Columbia.
The most significant lessons learned from these events were:
- Engage relevant organizations quickly.
- Know what information can and cannot be shared.
- Share the right information with the right people.
- Nurture strong, collegial relationships between key players for fast, effective collaboration.
Mechanisms in place to identify priorities
Canada has a number of mechanisms in place to identify priorities for national non-enteric zoonotic disease surveillance and control. These include public health governance structures, programmatic exercises (environmental scans), and informal technical working groups of federal, provincial and territorial experts.
Public health governance structures
Zoonotic diseases can be brought forward for discussion and prioritization (as required), to a number of public health governance structures and informal working groups, including:
- the Council of Chief Medical Officers of Health,
- the Communicable and Infectious Disease Steering Committee,
- the Public Health Network Council,
- the Council of Chief Veterinary Officers,
- the National Farmed Animal Health and Welfare Council,
- PHAC/CFIA Non-Enteric Zoonotic Diseases Steering Committee, and
- Federal Partners in Wildlife Health.
Programmatic exercises
In 2012, PHAC conducted an assessment of non-enteric zoonotic diseases of public health significance with the highest risk of emergence or re-emergence in Canada. Risks were identified according to internationally accepted indices defining disease emergence and other criteria, including:
- Increased incidence in humans
- Increased geographic range
- Status of surveillance systems
- Necessity for inter-sectoral collaboration (between human public health and veterinary public health) in each province and territory
- Necessity for national collaboration in research, surveillance, diagnostic testing or communication and education
This assessment identified four disease priorities:
- Lyme disease and other emerging tick-borne diseases [highest score]
- West Nile virus and other emerging mosquito-borne diseases
- Rabies
- Zoonotic influenza
The findings were finalized and presented to the Communicable and Infectious Diseases Steering Committee of the Public Health Network Council in 2013. They were used to inform areas of future federal, provincial and territorial collaboration on non-enteric zoonoses in Canada.
In addition, the Infectious Diseases Prevention and Control Branch does annual strategic and operational planning exercises to identify emerging risks and develop action plans to address them, such as the Zika virus action.
Technical working groups
Two communities of practice, or informal technical working groups: the West Nile Virus and Other Mosquito-Borne Disease Surveillance Working Group, and the Lyme and Other Tick-borne Disease Surveillance Working Group-also bring forward recommendations on zoonotic disease issues for discussion and prioritization to a number of committees and councils in Canada, including:
- Communicable and Infectious Diseases Steering Committee of the PHN
- Council of Chief Medical Officers of Health of Canada
- Council of Chief Veterinary Officers
- PHAC-CFIA Non-Enteric Zoonotic Diseases Steering Committee
- Federal Partners in Wildlife Health Director General Committee
Laboratory capacity for zoonoses and animal health
In Canada, public health and animal health laboratories are often housed together. However, they are not shared laboratories. There are several mechanisms in place for sharing data and information between human and animal laboratories when an urgent issue requires a collaborative approach. Although there are processes in place for sharing information, specimens, data and reports between public health and animal health laboratories as part of regular business, this information is not necessarily shared systematically as livestock laboratory results and human diagnostic test results are usually confidential.
Animal population surveillance systems
The Canadian Food Inspection Agency is the primary federal organization dealing with reportable animal diseases in Canada of significance to human or animal health or to the Canadian economy. The CFIA's surveillance work is supported by the Canadian Animal Health Surveillance Network and the Canadian Animal Health Surveillance System. Both are networks of animal health diagnostic laboratories and surveillance activities created to help integrate animal health surveillance data across multiple jurisdictions.
The Canadian Wildlife Health Cooperative is a node of the Canadian Animal Health Surveillance System. Its regional centres are located in Canada's veterinary colleges and animal health centres. It maintains a centralized national repository for wildlife health data, including diagnostic and testing data, as well as observational data (e.g. citizen science) and other external sources of data.
The Cooperative generates situational awareness and reliable assessments for federal departments on the state of wildlife health in Canada. The Cooperative and the U.S. Geological Survey's National Wildlife Health Center are a joint World Organization for Animal Health collaborating centre, with expertise in various fields of wildlife health and disease.
The Public Health Agency of Canada collects data on the incidence of food-borne illness, including those of animal origin, through its National Enteric Surveillance Program. In collaboration with provincial and territorial partners, PHAC also conducts animal surveillance for non-enteric zoonotic diseases of public health significance. This includes national surveillance of ticks and Lyme disease, surveillance of West Nile virus, and rabies surveillance in livestock and wildlife.
During West Nile virus season, for example, the CFIA shares reports with PHAC on data from domestic animals (such as horses) testing positive for West Nile virus. This data, along with other indicators, allows local and provincial public health officials to assess the current risk to humans from West Nile virus.
PHAC and the CFIA co-fund collaborative surveillance activities with provinces, territories and NGOs for zoonotic diseases, such as avian influenza in waterfowl and shorebirds. In areas with endemic terrestrial rabies, provinces and territories monitor wildlife for rabies through their own laboratories and those of the CFIA. Most provinces have programs to monitor and manage priority zoonoses in animals, for example Quebec rabies control programs (Available in French only), Ontario Rabies in wildlife, New Brunswick Rabies Control Program and Nova Scotia Rabies Response Plan.
Privacy and mandate issues related to sharing data can affect the sharing of surveillance reports between public health and animal health laboratories. Common platforms such as the Canadian Network for Public Health Intelligence support the integration of many surveillance initiatives across human, animal, food, and environment domains.
In the event of a suspected zoonotic outbreak, Canada has two key mechanisms in place to ensure collaboration and information sharing between human and animal health sectors at the federal level: PHAC's Health Portfolio Operations Centre and the CFIA's National Emergency Operations Centre. All groups implicated in the response to an outbreak share reports and intelligence through these emergency operations centres via their supporting infrastructure and incident command systems.
For information on livestock populations, see the World Organization for Animal Health Performance of Veterinary Services Evaluation for Canada. Government departments at all levels, with a mandate for wildlife management and nature protection, monitor wildlife population status and trends in support of conservation goals.
Linkages between human and animal disease surveillance and outbreak response
Human and animal health programs are established in various federal, provincial and territorial government departments and agencies. However, depending on the requirements of the event, ministries of health, agriculture, wildlife, environment, natural resources, conservation and parks do collaborate on zoonotic disease detection, response and control, with the lead organization engaging others as appropriate.
Linkages are also created through a variety of governance structures, such as the Federal Partners in Wildlife Health Director Generals' Steering Committee, which brings together Environment and Climate Change Canada, PHAC, the CFIA and other partners. Committee members collaborate on wildlife health and disease issues; set federal goals and program priorities; and provide federal input to and coordinate funding for the Canadian Wildlife Health Cooperative-a key partner in wildlife surveillance, assessment, investigation and response in Canada.
Canada is developing a National Wildlife Health Strategy that describes the country's vision for wildlife health and identifies the challenges, opportunities and actions involved in achieving that vision. A working group of federal partners is collaborating with the Canadian Wildlife Health Cooperative and provinces and territories on the Strategy.
Canada has a Plant and Animal Health Strategy that sets out a vision for Canada consistent with One Health principles. It describes how multiple organizations will coordinate their activities to safeguard plant and animal health in Canada. Wildlife considerations are included in the strategy primarily in relation to the interface between managed and unmanaged populations.
Public reporting and information sharing
In Canada, situational awareness reports and reports of new, possible and ongoing zoonotic disease threats are routinely shared between agencies through working groups established to manage specific issues, such as avian influenza and chronic wasting disease. These reports are posted on the Canadian Wildlife Health Cooperative website.
More recently, reports on potential new issues and updates on existing issues are shared between agencies through the PHAC-CFIA Non-Enteric Zoonotic Disease Steering Committee. The situational awareness report on the first recorded human case of locally-acquired eastern equine encephalitis in Ontario in late 2016 is an example of inter-agency collaboration for reporting and information sharing.
In early November 2016, the CFIA alerted PHAC to a bovine tuberculosis investigation in Alberta and Saskatchewan. Since then, regular updates on the situation have been shared through the Council of Chief Veterinary Officers calls, bilateral technical exchanges, emergency operations centres, and public affairs communications activities. In addition, PHAC is working with provincial agriculture and health personnel to monitor for possible human exposures or cases.
PHAC and the CFIA have collaborated on assessing the risks associated with animal importation, such as monkeypox in animals from endemic areas of Africa; and on responses to outbreaks of concern to animal health, animal welfare, and human health, such as pandemic influenza pH1N1 in 2009.
P.4.2 Veterinary or animal health workforce
Graduate-level training
There are five veterinary colleges in Canada with active research and teaching programs and that operate veterinary diagnostic laboratories: the Faculty of Veterinary Medicine, University of Calgary; the Ontario Veterinary College, University of Guelph; the Atlantic Veterinary College, University of Prince Edward Island; the Faculté de médecine vétérinaire (Available in French only), Université de Montréal; and the Western College of Veterinary Medicine, University of Saskatchewan.
Canadian veterinary colleges are accredited by the American Veterinary Medical Association Council on Education. Through this accreditation, all veterinarians graduating from accredited Canadian schools are also accredited by the Royal College of Veterinary Surgeons of the United Kingdom.
Veterinary medicine courses in these institutions are based on international best practice and they reference the World Organization of Animal Health "Day 1 graduate" competencies. Veterinarians may become certified in a number of specialties, such as toxicology, veterinary microbiology, veterinary pathology and veterinary preventive medicine (with subspecialties in epidemiology, food safety and veterinary public health).
The minimum professional standard for practicing veterinary medicine in Canada is graduation from a recognized school of veterinary medicine, a certificate of qualification from the National Examining Board of the Canadian Veterinary Medical Association and appropriate provincial or territorial licensure.
Supplemental veterinary public health training opportunities
Veterinarians with degrees in public health and epidemiology at the graduate level are eligible to participate in PHAC's Canadian Field Epidemiology Program. A small number of veterinarians have participated in the program as either students or teachers since the program's inception in 1975.
PHAC and the CFIA are currently working to build Canada's veterinary epidemiology capacity through on-the-job training opportunities, such as hiring veterinary epidemiology students and recent graduates, Agency interchanges, and initiating collaborative projects.
Several provincial ministries of health have engaged veterinary public health epidemiologists in their infectious disease programs, including British Columbia's Centre for Disease Control, Manitoba Health Seniors and Active Living, and the Ontario Ministry of Health and Long-term Care.
P.4.3 Mechanisms for responding to infectious and potential zoonotic diseases are established and functional
National strategies for zoonotic outbreaks
Canada's Public Health Response Plan for Biological Events (2017) covers a range of biological public health events, including zoonotic disease outbreaks. It lays out a scalable governance framework for a coordinated health sector response to large-scale, multi-jurisdictional domestic events and international events, such as pandemic influenza, Zika virus, and Ebola virus, which require national coordination.
Canada also has a number of disease-specific plans and strategies in place, which lay out how various sectors collaborate during a response to a zoonotic disease outbreak, including:
- North American Plan for Animal and Pandemic Influenza
- Canadian Rabies Management Plan, 2009 (currently being updated)
- Federal Framework on Lyme Disease, 2017
- Food-borne Illness Outbreak Response Protocol, 2010 (guides how federal, provincial and territorial public health and food safety authorities manage food-borne illness outbreaks)
Examples of coordination between the sectors
Canada's National Plant and Animal Health Strategy, 2017 was developed in collaboration with industry, NGO, academic and government partners.
Health Canada, the PHAC and the CFIA have a Memorandum of Understanding for Common Issues Related to Human Health (2008). The Agencies have signed a Letter of Agreement Regarding Zoonotic Surveillance and Risk Assessment Process (2008). Recent zoonotic events that required a coordinated response across different sectors and levels of government include:
- Seoul virus (2017)
- Zika virus (2016/17)
- Bovine tuberculosis (2016)
- First recorded human case of locally-acquired eastern equine encephalitis (2016)
- Influenza [H3N2v (2016); H5N2 - low path (2016); H5N2 HPAI (2015); H5N2 HPAI (2014)]
- Ebola virus (2015/2016)
- Salmonella in live chicks (2015)
- Chikungunya (2014)
During these events, and consistent with the scale of the event, information was shared within the Health Portfolio and with other federal, provincial and territorial partners, international stakeholders, and non-government partners. Information sharing was done through regular meetings and calls as part of the emergency operations centre's business cycle for the event and through the responsible program area as appropriate.
During the response to Zika, Canada shared information on numbers of cases with the World Health Organization through the Pan-American Health Organization (PAHO), following International Health Regulation (IHR) procedures. During Ebola, a working group made up of representatives of different sectors produced guidance documents, which were shared on-line, including guidance for veterinarians provided by the Canadian Public Health Association.
During an event, sectors meet regularly but the frequency of meetings depends on the needs of the specific event-the severity, impact and how quickly the situation evolves. Roles and responsibilities for zoonotic disease outbreak response and control depend on the disease under investigation and whether it is reportable or notifiable in animal and human health at the provincial and territorial or federal level. In general, if the disease is not reportable in animals, then the responsibility to investigate and implement public health actions often falls to public health authorities.
During the response to Salmonella in live chicks, local public health authorities handled the human health investigation of cases. Provincial public health authorities coordinated the investigation within each respective province; and PHAC coordinated the investigation overall. The animal health investigation was led by the provincial agriculture authority where the hatchery operated with support from affected provinces.
Provincial public health coordinated the overall investigation into the Seoul virus outbreak (which affected only one province), while local public health conducted the human health investigation of the cases. Provincial agriculture helped develop testing guidelines, public communications, and engaged the veterinary community. PHAC communicated with key international stakeholders, the WHO/PAHO and other IHR National Focal Points and the U.S. Centers for Disease Control and Prevention. The National Microbiology Laboratory, the only laboratory in Canada with the required expertise, provided diagnostic support.
The CFIA led the response to avian influenza and also played a primary role in disease control on affected farms. Local and provincial public health conducted the human health investigations, while PHAC supported the CFIA and provincial public health authorities as needed.
Best practices, challenges, gaps and recommendations
Canada has robust systems in place at all levels of government to prevent, detect and respond to enteric and non-enteric zoonotic disease outbreaks. (For details on enteric zoonoses, see section P5: Food safety.)
There are many well-managed animal and human surveillance systems in place at all levels in Canada to monitor for emerging and re-emerging zoonotic diseases with the potential to affect human populations. Canada can improve its capacity to integrate the animal and human health sectors, particularly when it comes to timely sharing of surveillance data and collaboration on response and control activities.
One Health principles are generally accepted in Canada as key to understanding and managing zoonoses. The integration of human, animal and environmental factors is often considered and applied in policy and program decision making and in the response to rapidly changing disease dynamics at the interface of those factors.
Although Canada does not have a national One Health strategy, One Health ideas have been integrated into several national strategies, including the draft National Wildlife Health Strategy and the National Plant and Animal Health Strategy. Moreover, many public health governance structures and technical or policy working groups that deal with zoonoses follow One Health principles.
Canada has surveillance systems, plans, decision-making algorithms, protocols and partnership agreements that guide and support the response to zoonotic disease outbreaks. The Food-borne Illness Outbreak Response Protocol covers enteric zoonoses, while several disease-specific response plans support outbreaks of non-enteric zoonoses. As well, Canada's Public Health Response Plan for Biological Events could support the response to a large-scale zoonotic disease event.
The spread of infectious disease from animals to humans presents unique challenges that include complex scientific assessments, information sharing across sectors and jurisdictions, and the involvement of diverse groups of experts from multiple cross-sectoral organizations. While Canada has done significant work to build its capacity to respond to zoonotic disease events, there are still steps that could be taken to move closer to a comprehensive One Health approach, including:
- Continue to strengthen the integration of animal and human surveillance and reporting systems.
- Enhance response plans to include clear roles and responsibilities in the coordination, investigation and response to multi-disciplinary, multi-jurisdictional zoonotic disease events.
- Establish informal national working groups to address issues related to priority non-enteric zoonotic diseases.
P5: Food safety
Joint external evaluation target: States parties should have surveillance and response capacity for food and water borne disease risk or events. It requires effective communication and collaboration among the sectors responsible for food safety and safe water and sanitation.
Level of capability in Canada
Canada has a strong food safety system for the prevention and control of food-borne illnesses. This system is implemented through regulatory, public health, and risk analysis efforts across the farm-to-table continuum. Canada's federal food safety system is built on internationally recognized standards and a risk-based inspection approach. Recognized in the 2014 World Ranking Food Safety Performance report, as an overall leader in food safety performance, Canada is consistent in setting best practice standards in these areas.
Government responsibility for food safety and outbreak response is shared among federal, provincial and territorial governments. Canada's ability to anticipate, mitigate and respond to food safety risks requires the cooperation of federal partners, health professionals, local health authorities, and provincial and territorial ministries of health and agriculture.
Government has the authority to establish regulatory standards, inspect facilities, and act to address issues of concern, but industry has the primary responsibility to ensure that food products are safe and meet regulatory requirements.
Canada's capacity to protect the population from unsafe food has grown through improvements to enteric disease surveillance and laboratory diagnostic programs, formal outbreak response protocols and new regulatory approaches. In 2012 the federal government enacted the Safe Food for Canadians Act, which consolidates the authorities of the Fish Inspection Act, the Canada Agricultural Products Act, the Meat Inspection Act, and the food provisions of the Consumer Packaging and Labelling Act. The new act came into force at the same time as the Safe Food for Canadians Regulations and aims to:
- Make food as safe as possible for Canadian families
- Protect consumers by targeting unsafe practices
- Implement tougher penalties for activities that put health and safety at risk
- Provide better control over imports
- Institute a more consistent inspection regime across all food commodities
- Strengthen food traceability
Canada's capacity to detect and respond to food-borne outbreaks has also improved dramatically in the past two decades. Since 1996, the Public Health Agency of Canada (PHAC) has used the PulseNet Canada network of laboratories to "fingerprint" the DNA of bacteria to enhance outbreak detection and define the scope and scale of outbreaks beyond traditional methods.
Following an outbreak of listeriosis in 2008, Canada invested significantly in building its inspection and risk assessment capacity through Renewal of Government Response and Action Plan to the 2008 Listeriosis Outbreak, Food Safety Modernization and Food Safety Oversight.
Indicators
P.5.1 Mechanisms are established and functioning for detecting and responding to food-borne disease and food contamination.
Roles and responsibilities for national food safety and outbreak response
The federal, provincial and territorial governments in Canada share responsibility for food safety and public health. Provincial and territorial public health authorities have jurisdiction under their own authorities for foods produced and sold within the province or territory. The federal government establishes national standards and policies for and addresses issues with multi-jurisdictional and international or trade implications.
Health Canada (Food Directorate) sets standards and policies, which are aligned with the Codex Alimentarius Commission where appropriate, for the safety and nutritional quality of food sold in Canada. Health Canada also:
- Conducts health risk assessments for food-borne hazards
- Manages the safety of veterinary drugs in food-producing animals
- Provides reference services (botulism) and expertise, including expertise in chemical food contamination (for more information on how Canada manages chemical contamination, see section Chemical events below)
Health Canada's Bureau of Microbial Hazards is an International Food Safety Authorities Network (INFOSAN) Focal Point. Health Canada is also responsible for informing Canadians about potential risks to their health.
The Canadian Food Inspection Agency is responsible for:
- Delivering all federal food inspection programs
- Monitoring industry's compliance with food-related acts and regulations
- Conducting food safety investigations (trace back)
- Undertaking enforcement actions, or requesting industry to recall food, as necessary
- Communicating with the public about potential food safety risks
Three groups within the Canadian Food Inspection Agency play key roles in the response to food-borne illness outbreaks:
- Regional inspection staff, including Area Recall Coordinators and Regional Recall Coordinators, conducts food safety inspection activities. Area Recall Coordinators are the usual first point of contact for local or regional health units, provincial and territorial governments, and companies when they identify an issue with their food product.
- The Office of Food Safety and Recall, which coordinates food safety investigations and is responsible for the consistency of decision-making on food recalls. The Office of Food Safety and Recall is the usual first point of contact for national and international food safety related issues and the INFOSAN emergency contact point for Canada. The Office links with Health Canada for health risk assessments and with PHAC for food-borne illness outbreak investigations.
- The Food Safety Science Directorate is responsible for providing scientific analysis and guidance to Canadian Food Inspection Agency staff and provincial and territorial partners.
The Canadian Food Inspection Agency establishes policies and standards for the import of foodstuffs, live animals and animal products, and the Canada Border Services Agency enforces these policies and standards. The Canada Border Services Agency has the legislated power to undertake inspections, seizures, testing, holding, destruction or rejection of products, including live animals, at ports of entry.
The Public Health Agency of Canada plays a leadership role in coordinating the response to multi-jurisdictional food-borne illness outbreaks (outbreaks that occur in more than one province, territory, or country). The Agency also provides support to provinces and territories responding to outbreaks within their jurisdictions when assistance is requested.
PHAC's Infectious Disease Prevention and Control Branch assesses the risk and reduces the impact in Canada and internationally of infectious diseases that can be spread to humans through contaminated food or water, or through contact with infected animals or the environment. The Branch works closely with provincial, territorial and local health departments to conduct ongoing epidemiological disease surveillance for enteric illnesses.
The Branch is the first point of contact for the federal government in managing issues related to food-borne illness outbreaks and an INFOSAN Focal Point. The Branch leads epidemiological and laboratory investigations related to multi-jurisdictional outbreaks. It does this in partnership with Health Canada and the Canadian Food Inspection Agency, and following established protocols. The Branch communicates with Canadians about health risks and how to prevent illness during an outbreak.
PHAC's National Microbiology Laboratory provides reference services for strain identification and characterization, national laboratory-based surveillance, and dissemination of information through PulseNet Canada. This is done through the National Enteric Surveillance Program.
The National Microbiology Laboratory, through PulseNet Canada, is the first point of contact for provinces and territories sharing strain identification data and the detection of clusters of strains that are occurring in more than one jurisdiction-which would identify the potential for multi-jurisdictional food-borne outbreaks. Health Canada provides reference services for botulism, listeriosis and vibrio (in partnership with the National Microbiology Laboratory), as well as reference services for viruses in food.
Canada has a number of mechanisms in place to identify national food safety priorities, including committees within the Public Health Network (described in section P4: Zoonotic disease above). In addition the Federal, Provincial and Territorial Food Safety Committee coordinates the development of national food safety policy options, implements initiatives to achieve national goals, and sets priorities to enhance accountability.
Provinces and territories have their own food safety legislation in place and conduct local epidemiological investigations; inspections of food produced and sold in their jurisdiction; and education activities to reduce risks related to food. They share responsibility with the federal government for inspection of food traded within their jurisdiction. Provincial, territorial and local health departments have the primary statutory authority for enteric disease surveillance in their jurisdiction.
Most food-borne disease outbreaks are local events in just one city and local public health officials investigate. Provincial and territorial health departments have their own outbreak response protocols and may work with their departments of agriculture and the local public health community to guide a coordinated response to food-borne illness outbreaks within their jurisdiction. Provinces and territories may request help from federal departments, such as Health Canada, Public Health Agency of Canada, and the Canadian Food Inspection Agency to respond to an emergency or conduct an investigation.
Industry is responsible for the production of safe food in compliance with all government standards and laws, and for conducting food recalls when required.
Specialized training and resources for food-borne outbreak investigation
PHAC, the Canadian Food Inspection Agency, Health Canada and the provinces and territories have trained resources with experience in food-borne outbreak investigation. These include epidemiologists, microbiologists, medical doctors, veterinarians and other public health professionals (e.g. public health inspectors, public health nurses). At the federal level, all new food inspectors undergo training on their roles and their responsibility to deliver the programs under the Safe Food for Canadians Act. Several PHAC epidemiologists are also trained in food facility investigations and product tracing. In Canada, these trained, experienced people at the national level take part in outbreak response teams.
PHAC's Field Service Training and Response division provides outbreak investigation training through a number of programs, including: Surveillance and Outbreak Core Skills, the Canadian Public Health Service, and the Canadian Field Epidemiology Program.
Food-borne disease outbreak investigations in Canada follow standard epidemiological principles. PHAC is the federal lead for coordinating multi-jurisdictional outbreak investigations, public communications on health risks associated with specific outbreaks, and the steps people can take to protect themselves. The Archived: Food-borne Illness Outbreak Response Protocol guides multi-jurisdictional collaboration in response to food-borne illness outbreaks in Canada by setting out guiding principles and operating procedures to improve the efficiency and effectiveness of response.
PHAC often shares investigation-specific questionnaires for multi-jurisdictional outbreaks with affected provinces and territories to ensure standard data collection and analysis. In some events, investigators will conduct case interviews centrally, using a single interviewer approach. To coordinate a multi-agency response to a food-borne illness outbreak that affects several provinces or territories, Canada establishes an Outbreak Investigation Coordinating Committee with representatives from Health Canada, the Canadian Food Inspection Agency, PHAC and the affected provinces or territories. The Committee assesses information requirements and identifies the partners best suited to gather the required information. Every effort is made to standardize the information collected. Typical investigation steps include:
- PHAC, along with public health staff in the affected provinces and territories and other Committee members, as appropriate, determines what the case definition will be for the investigation.
- PHAC investigators centrally collate and analyze case information (e.g. line lists and case questionnaires) using the information sent to them from provincial and territorial public health partners.
- Findings from the epidemiological, laboratory and food safety investigations are shared with the partner members of the Committee and integrated to identify the potential cause and source of the outbreak and areas for further investigation.
- The local public health units and healthcare oversee collection of appropriate clinical specimens in accordance with infectious disease protocols and laboratory guidelines. The Food-borne Illness Outbreak Response Protocol outlines the standard operating procedure for directing food samples collected during epidemiological, public health, and food safety investigations to the federal laboratory network.
- Led by PHAC's National Microbiology Laboratory, PulseNet Canada is a real-time molecular subtyping network for food-borne disease surveillance supporting the rapid detection and response to single- and multi-jurisdictional outbreaks. PulseNet Canada also provides laboratory investigation during multi-jurisdictional outbreak response and support for single jurisdiction response to enable timely identification of outbreak sources and associated public health action. The work of PulseNet Canada has been critical for the detection and response to large national outbreaks.
Coordination, communication, and collaboration among stakeholders
Federal, provincial and territorial governments work together when there is a national or international outbreak of food-borne illness. They rely on the following tools to ensure effective collaboration:
- The Food-borne Illness Outbreak Response Protocol is the technical and operational protocol that guides how federal, provincial and territorial public health and food safety authorities work together to investigate and manage national or international food-borne illness outbreaks. The Protocol establishes clear lines of communication among all partners and stakeholders and is periodically reviewed and shared with partners and appropriate public health professionals for their endorsement.
- The Multi-Lateral Information Sharing Agreement governs the uses and disclosure of information during public health events and has a specific annex covering Public Health Emergencies of International Concern.
- The interaction between Health Canada and the Canadian Food Inspection Agency during an event or emergency is detailed in a 1999 memorandum of understanding and its related framework, Roles and Responsibilities Framework for Federal Food Safety and Inspection Activities (June 1999).
- A 2008 trilateral memorandum of understanding between Health Canada, PHAC and the Canadian Food Inspection Agency describes the roles and responsibilities regarding common issues that impact human health. These issues include food safety and nutrition, infectious disease outbreak management and emerging zoonotic diseases.
- PHAC, the Canadian Food Inspection Agency and Health Canada have developed the Federal Food Safety Communications Protocol for National Food-Borne Illness Outbreaks (shared in 2015), and a joint protocol for sharing information between Health Portfolio and international partners, including foreign food safety authorities, INFOSAN and the WHO during an event with international implications. (See D3: Reporting)
- The Canadian Food Inspection Agency's Protocol for Sharing of Information during Food Safety Investigations and Recalls describes the type of information that can be shared with other government departments, other countries, regulated parties, third parties and the public during food safety investigations and recalls. It also describes how information is shared and the type of information that the Canadian Food Inspection Agency is obligated to protect. The protocol describes how Canada shares information with other countries to enable follow up action.
PHAC's National Microbiology Laboratory coordinates the collection and centralized analysis of all laboratory data via the PulseNet Canada network. This network is comprised of provincial public health laboratories and federal food safety partners. The National Microbiology Laboratory provides the analyses and interpretations of laboratory data for Outbreak Investigation Coordination Committee discussions.
Federal, provincial and territorial food safety and epidemiology investigators participate in Outbreak Investigation Coordination Committee discussions at which they share information required to mitigate or contain the impact of the outbreak in a timely and effective way. During an event, PHAC, the Canadian Food Inspection Agency and Health Canada have regular conference calls as part of the coordination of information sharing related to new and ongoing events.
The public health and food safety organizations participating in an Outbreak Investigation Coordination Committee each have separate mandates and responsibilities to communicate with stakeholders within their respective portfolios. For example, with respect to routine surveillance, PHAC programs, such as the National Enteric Surveillance Program and FoodNet Canada, routinely communicate with the public health and food safety organizations which represent the farm-to-fork continuum.
The Canadian Food Safety Information Network is a collaborative national (federal/provincial/territorial) network that will strengthen the capacity of food safety authorities across the country to anticipate, detect and respond to food safety events through timely information sharing and coordinated action.
As INFOSAN Focal Points PHAC and Health Canada have close links to international food safety authorities and share information routinely and during emergencies. Canada also has two members on the INFOSAN Advisory Group established in 2006.
PHAC works collaboratively with Health Canada, the Canadian Food Inspection Agency, and with provincial and territorial counterparts to determine risks to the health of the Canadian public associated with food-borne illnesses. The Agency leads public communication on how to prevent food-borne illness during a multi-jurisdictional food-borne illness outbreak. The Agency is also the International Health Regulations (2005) Focal Point (see P2: International Health Regulations coordination, communication and advocacy for more information).
Canada works with several international partners to deal effectively with the international aspect of many food safety issues, and to foster international collaboration. These partners include:
- The U.S. Food and Drug Administration, as part of the Joint Action Plan for the Canada-United States Regulatory Cooperation Council, to develop an agreement to facilitate trade between the two countries based on the assurance that appropriate measures are in place to prevent food contamination and food-borne illness and to detect and respond in the case of an event.
- The U.S. Centers for Disease Control and Prevention and the U.S. Food and Drug Administration to manage and control food-borne illness outbreaks and to enable sharing of laboratory surveillance data in real-time through PulseNet, including whole genome sequence data. PHAC's National Microbiology Laboratory is also a member of the PulseNet International Steering Committee, providing strategic guidance and technical expertise to the international PulseNet network that comprises 86 countries.
- The Asia Pacific Economic Cooperation Food Safety Forum and the Global Food Safety Partnership, to provide technical expertise and to help build global outbreak response capacity.
- WHO (Global Foodborne Infections Network, Foodborne Epidemiology Reference Group, and the Advisory Group on Integrated Surveillance of Antimicrobial Resistance), Pan American Health Organization, Codex Alimentarius, and Food and Agriculture Organization's Working Group to Develop a Handbook for Food Safety Risk Communications. PHAC shares technical and scientific expertise and risk communications advice and has worked on technical papers and guidance documents, such as Applications of Whole Genome Sequencing in food safety management and Guidance for the use of whole genome sequencing in developing countries.
- The Global Health Security Action Group, Communicators Network, with public health and emergency preparedness communicators from the G8 countries plus Mexico, as well as from the WHO and the European Centre for Disease Prevention and Control. PHAC shares communications plans and messages with its international partners, when appropriate, to help foster consistent and coordinated international communications.
- The INFOSAN secretariat, to create and deliver a technical training series for INFOSAN members on food-borne outbreak investigations, food safety investigations, health risk assessment and food recalls, food-borne disease surveillance, and the Canadian Food Inspection Agency protocol for notification of international partners.
Multi-sectoral risk profiling and risk management
Food-borne Illness Outbreak Response Protocol procedures focus on a coordinated response to food-borne illness outbreaks. They do not specifically address the broader risk assessment process that contributes to policy development and standard-setting to reduce the risk of future outbreaks. However, the Protocol's event review process provides an opportunity to capture lessons learned and make recommendations for future policy development to manage risk.
With respect to routine ongoing surveillance, PHAC plays a role in leading surveillance programs that support outbreak detection and providing a more integrated approach to identify and address public health risks in the food chain. The Agency is engaged in cutting-edge science activities, including the development of laboratory technology to enhance the ability to detect and characterize food-borne illness outbreaks and their sources.
PHAC is the lead on the following four ongoing surveillance programs that allow federal, provincial, territorial and local public health authorities to monitor trends, identify potential outbreaks or events, and implement risk management and response strategies:
- The National Enteric Surveillance Program supports timely detection of outbreaks through weekly monitoring of enteric pathogens reported in Canada and provides timely analysis and reporting of laboratory confirmed enteric disease cases in Canada. Program data is used to determine if reductions in food-borne illness are observed over time.
- PulseNet Canada provides high resolution outbreak detection on a daily and weekly basis. Strain-level data on each case of bacterial food-borne illness are analyzed and reported daily and weekly to detect multi-jurisdictional outbreaks. It also provides all data collection analysis during outbreak response.
- The Enhanced National Listeriosis Surveillance Program collects detailed clinical, demographic, and risk factor information on invasive listeriosis cases in participating provinces and territories in order to inform surveillance, research, and outbreak investigations related to invasive listeriosis.
- The FoodNet Canada surveillance program is an integrated food safety surveillance system that protects the health of Canadians by identifying the causes of human illness. By collecting samples from local farms, grocery stores, and water sources and linking this information with human illness, FoodNet Canada can identify the food sources causing illness by region and across Canada. This provides practical public health risk information to the federal, provincial and territorial governments, industry, and national food safety partners. This information is used to enable targeted prevention, prioritize risk mitigation and oversight, and provide evidence on the effectiveness of the food safety system.
PHAC plays a leadership role in public health research along the food continuum to target areas that will improve public health outcomes. Public health research at the molecular and population levels (using methods such as genomics, lab based studies, epidemiological studies, risk modelling, decision analysis, risk mapping and knowledge synthesis) are used to focus on the prevention of public health risks. These risks arise from the food chain, animals and the environment. Information from this research provides the evidence for understanding risks to health and potential control strategies.
It is critical to understand the burden of illness and its associated public health impact and cost, beyond what is captured through laboratory based surveillance. This knowledge is central to measuring the performance of the Canadian food safety system and supporting food safety decision making. Drawing from Canada's multiple complementary food-borne disease surveillance systems, PHAC estimates that each year about one in eight Canadians (four million people) get sick from the food they eat. This accounts for over 11,500 hospitalizations and 240 deaths each year due to food-related illness.
Communication with the public about food safety and food hazards
Provincial and territorial food safety authorities across Canada recognize the importance of effective communications and have dedicated resources in place. Provinces and territories make food safety information, including alerts and advisories, publicly available online. British Columbia's Food Safety & Security site, for example, includes links to food recalls and allergy alerts and public awareness materials, such as "Easy Ways to Make Food Safe." New Brunswick's Food Safety Resources site includes fact sheets, posters and tools, as well as links to federal guidance document and websites.
Canada's Food and Nutrition portal is the public gateway to food safety information from federal government departments and agencies. Some sections of the website allow subscribers to sign up for automatic notifications when information is posted about food recalls and outbreaks.
- Food recalls and alerts
- Canadian Food Inspection Agency food safety investigations
- Public health notices
Authorities that establish Canada's food management systems
The Canadian Food Inspection Agency exercises its food safety mandate under the following authorities. Although not exhaustive, the list illustrates the broad authority of the Canadian Food Inspection Agency and its responsibility in Canada for the whole food continuum:
- Canadian Food Inspection Agency Act
- Food and Drugs Act
- Safe Food for Canadians Act
- Health of Animals Act
- Canada Agricultural Products Act
- Consumer Packaging and Labelling Act
- Feeds Act
- Fertilizers Act
- Fish Inspection Act
- Meat Inspection Act
- Plant Protection Act
- Seeds Act
Health Canada and PHAC exercise their food safety mandates under the Food and Drugs Act, as well as the Department of Health Act and the Public Health Agency of Canada Act, respectively.
Examples of recent food safety actions
In accordance with the Food-borne Illness Outbreak Response Protocol, PHAC leads multi-jurisdictional outbreaks affecting more than one province or territory. Between April 1, 2016 and March 31, 2017, the Agency led nine multi-jurisdictional outbreak investigations-compared to 10 in the previous fiscal year. Descriptions of many of these food-related outbreaks are available on the Agency website, including:
- Outbreak of E. coli infections linked to various flours and flour products, 2017
- Outbreak of norovirus and gastrointestinal illnesses linked to raw and undercooked oysters, 2017
- Outbreak of hepatitis A infections linked to frozen fruit product, 2016
- Outbreak of Listeria infections linked to packaged salad products, 2016
- Archived: Outbreak of Salmonella infections linked to chia and flax seed powder, 2016
- Outbreak of Vibrio parahaemolyticus linked to raw shellfish, 2015
Recent food recalls and investigations include:
- Archived: Salmonella in frozen uncooked breaded chicken, October 2017
- Archived: Marine bio toxin in oysters, October 2017
- Archived: Norovirus in raspberry mousse cake, August 2017
- Archived: Undeclared milk (allergen) in Chinese sausages, September 2017
- Archived: Undeclared gluten (allergen) in infant cereal, September 2017
- Archived: Undeclared sulphites (allergen) in dried and preserved products, August 2017
Reviews and evaluations are essential components of the food safety system in Canada. After a large outbreak or exercise involving multiple stakeholders, the lead agency chairs an "after-action review." This is conducted with appropriate participating partners to produce an after-action report, which informs a capabilities improvement process. For example, following large E. coli events, the Government of Canada developed policy on labelling mechanically tenderized beef, and guidance on E. coli 0157:H7 and E. coli 0157:NM in raw beef.
The Canadian Food Inspection Agency's Program Management Framework is designed to be used proactively and reactively to identify and address food safety risks and performance issues. The Framework was recently used to review and improve performance in the wake of outbreaks associated with E. coli in flour and Salmonella in raw breaded chicken products.
In cases of very serious outbreaks, the Government of Canada will appoint an independent investigator to review the event and make recommendations. For example, following a 2008 Listeria outbreak, the Weatherill Report proposed 57 recommendations to improve Canada's food safety system. The Government of Canada's response to the recommendations was also published online.
Best practices, challenges, gaps and recommendations
Canada has a strong food safety system for the prevention and control of food-borne illnesses. Federal partners work closely with each other as well as with provincial and territorial and local agencies, industry and industry associations, and consumers.
Formalized multi-agency and multidisciplinary participation through every stage of detecting, investigating, and responding to food-borne outbreaks and illness has become routine during multi-jurisdictional outbreak investigations, yet challenges still remain. Multi-jurisdictional outbreaks are difficult to detect and investigate due to the wide distribution and multiple sources of many food ingredients and products.
There are challenges related to timely collection of complete food histories, coordination of specialized laboratory testing to trace and track cases, the viability of viruses and parasites, and unexpectedly contaminated foods sources. Although coordination among federal agencies and experts from health, risk assessment and food sciences is consistent, there is a need to continuously evaluate, modernize, and strengthen the coordination of detection and investigation activities.
Developing new epidemiological tools that enhance food-borne illness outbreak investigations and reduce food-borne illnesses and deaths could help advance food safety capacity. It will be important for the Government of Canada to continue development of next-generation laboratory methods (such as whole genome sequencing) for pathogen identification.
Modernizing the PulseNet Canada network to capture and interpret genomic data is a high priority and is necessary to provide the best outbreak detection and response in Canada and to remain compatible with international partners. PHAC is in the process of transitioning all laboratory-based surveillance to use whole genome sequencing. There is also an opportunity to reinforce coordination and support for provinces and territories by conducting more clinical, food, and environmental testing, and isolate characterization.
P6: Biosafety and biosecurity
Joint external evaluation target: A whole-of-government national biosafety and biosecurity system is in place, ensuring that especially dangerous pathogens are identified, held, secured and monitored in a minimal number of facilities according to best practices; biological risk management training and educational outreach are conducted to promote a shared culture of responsibility, reduce dual use risks, mitigate biological proliferation and deliberate use threats, and ensure safe transfer of biological agents; and country-specific biosafety and biosecurity legislation, laboratory licensing, and pathogen control measures are in place as appropriate.
Level of capability in Canada
Canada has a national biosafety and biosecurity program for the oversight of activities with human and animal pathogens and toxins and regulated plant pests. The ultimate goal of the program is to reduce public health risks and potential risks to Canadian plant and animal resources posed by activities involving these materials.
Biosafety and biosecurity oversight requirements are predominately a federal responsibility, although requirements regarding worker safety, waste and accreditation of diagnostic laboratories exist within governments at the federal, provincial, territorial and municipal levels. Federal oversight has been developed with the objective of complementing existing federal, provincial and territorial regimes to reduce overall burden to regulators and regulated parties.
Indicators
P.6.1 Whole-of-Government biosafety and biosecurity system is in place for human, animal and agriculture facilities
Accountability for dangerous pathogens and toxins and regulated plant pests
Accountability for dangerous pathogens and toxins and regulated plant pests in Canada is a responsibility shared by federal, provincial and territorial partners. At the federal level, accountability is a collaborative effort between multiple departments including the Public Health Agency of Canada (PHAC), the Canadian Food Inspection Agency (CFIA), Global Affairs Canada, Environment and Climate Change Canada, Health Canada, Transport Canada, and the Canada Border Services Agency.
PHAC's Centre for Biosecurity is the national authority on biosafety and biosecurity for human pathogens and toxins and is responsible for their regulation under the authority of the Human Pathogens and Toxins Act and Human Pathogens and Toxins Regulations. PHAC is also responsible for the importation or transfer of terrestrial animal pathogens and toxins, with the exception of non-indigenous animal pathogens, emerging animal disease pathogens, and animal pathogens in animals, animal products, animal by-products, or other organisms, under the authority of the Health of Animals Act and regulations.
CFIA is the national expert on biosafety and biosecurity for foreign animal diseases and emerging animal diseases. It is responsible for regulating the importation or transfer of non-indigenous animal pathogens and emerging animal disease pathogens. This also extends to animals, animal products, and animal by-products that contain a terrestrial animal pathogen.
The Agency acts under the authority of the Health of Animals Act and the Health of Animals Regulations. It is also responsible for transfer and importation of aquatic animal and bee pathogens under these acts, and is Canada's national plant protection organization. It regulates the import and domestic movement of regulated plant pests and invasive plant species under the Plant Protection Act and Plant Protection Regulations, and the Seeds Act and Seeds Regulations.
Canada seeks to fulfil its obligations under the Biological and Toxins Weapon Convention through participation in the Australia Group. The Group is an informal forum that has developed harmonized export controls for human, animal, and plant pathogens and toxins with dual use potential. These controls also apply to dual use manufacturing facilities, equipment, technology, and software, as well as other items that could be used to test or disseminate controlled agents or used for protection against them.
In Canada, export controls have been implemented through Groups 2 and 7 of the Export Control List schedule. Global Affairs Canada administers export controls for strategic goods and technology under the authority of the Export and Import Permits Act. Residents of Canada wishing to export goods or technology listed on the Export Control List must have a Permit to Export issued by Global Affairs Canada.

Figure 3: Oversight of pathogens, toxins and regulated plant pests in Canada - Text description
Figure 3 illustrates the oversight of pathogens, toxins and regulated plant pests in Canada and highlights that it is a responsibility shared by a number of departments under the authority of various acts and regulations. Federal departments are depicted with red boxes and the relevant legislation is depicted by dark blue boxes.
Towards the top of the diagram is a long light blue box representing the Canadian border. On top of the box is the word "International". Above it is a box for Global Affairs Canada (GAC) with an arrow pointing to the right towards the Export and Import Permits Act, followed by an arrow pointing to the words "Toxicological and biological agents on the Export Control List", illustrating that GAC manages export controls for strategic goods and technology under this act.
On the Canadian border itself is a box for Canada Border Services Agency (CBSA) with an arrow pointing to the right towards the Canada Border Services Agency Act, indicating that CBSA is responsible for taking action to protect Canada's borders under this legislation, In this case, specifically monitoring the import and export of pathogens, toxins and regulated plant pests for federal partners.
Underneath the Canadian Border box is the word "National" and stemming from either side of the Canadian Border are dotted lines that go down and meet in the middle to form a box. This represents the oversight conducted domestically with respect to biosafety and biosecurity. On the left hand side of the dotted line is a box for Transport Canada, with an arrow pointing to the Transport of Dangerous Goods act and regulations. This describes Transport Canada's cross cutting role with respect to the safe and secure transport of pathogens, toxins and regulated plant pests.
In the centre of the diagram are the remaining departments with a key function in this area. These include Environment and Climate Change Canada (ECCC), Health Canada (HC), Canadian Food Inspection Agency (CFIA), and Public Health Agency of Canada (PHAC).
On the left side is a box representing ECCC and HC, with a purple arrow pointing down to a the Canadian Environmental Protection Act, 1999-New Substances Notification Regulations (Organisms). There is another purple arrow pointing down from there to the words New Substances (Organisms).
In the Centre is a box for CFIA with 2 yellow arrows pointing to 2 boxes. The first box "Plant Protection Act" has a yellow arrow leading to the word "Plant pathogens" and the second box "Health of Animals Act and regulations", has another yellow arrow pointing to the following list:
- Animal products which contain an animal pathogen
- Animal pathogens (Foreign Animal Diseases (FADs)/ Emerging Animal Diseases (EADs))
- Bee pathogens
- Aquatic animal pathogens
This illustrates CFIA's role in biosafety and biosecurity by overseeing laws governing plant pathogens, and some animal pathogens.
The final box on the right is for PHAC with 2 green arrows pointing to 2 boxes. The first box "Health of Animals Act and regulations" has a green arrow leading to the word "Terrestrial animal pathogens (pure culture), and the second box "Human Pathogens and Toxins Act and regulations", has another green arrow pointing to the words "Human pathogens and toxins". This highlights PHAC's responsibility with regards to both human and terrestrial animal pathogens and toxins, working within this legislative framework.
Environment and Climate Change Canada and Health Canada conduct environmental and indirect human health risk assessments, respectively, for new organisms in products regulated under the Food and Drugs Act (e.g., novel foods, human biologics and food additives) and recommend risk management measures.
Certain new living organisms, including microorganisms, proposed for importation into or production within Canada are subject to the Canadian Environmental Protection Act 1999 and the New Substances Notification Regulations (Organisms). A new substance, such as a microorganism developed through biotechnology, requires notification under the Canadian Environmental Protection Act 1999 prior to importation into or manufacture in Canada, if it is not already on the domestic substances list. This legislation protects both the environment and human health from potentially harmful animate biotechnology products.
Substances, including waste, containing pathogens classified as infectious, or toxins identified as toxic in transport are governed by the Transportation of Dangerous Goods Act and regulations, which is administered by Transport Canada.
The Canada Border Services Agency provides integrated border services that support national security and public safety priorities while facilitating the free flow of persons and goods. Under the Customs Act, border service officers have the authority to detain and examine any goods at the Canadian border. The Canada Border Services Agency also provides administrative support at Canadian points of entry for imported and exported pathogens and toxins and plant pests under the authorities of PHAC, the CFIA, and Transport Canada.
Provinces and territories may address other aspects linking to biosafety and biosecurity, including occupational health and safety, waste management and diagnostic accreditation.
National biosafety and biosecurity legislation, regulations, and guidelines
The Human Pathogens and Toxins Act and the Human Pathogens and Toxins Regulations, along with the Canadian Biosafety Standard, 2nd Edition, 2015, and the Canadian Biosafety Handbook, 2nd Edition, 2016, are pillars of Canada's safety and security program for human pathogens and toxins.
The Canadian Biosafety Standard is a harmonized national standard for the handling and storing of human and terrestrial animal pathogens and toxins in Canada. The Canadian Biosafety Handbook specifies the physical containment requirements, operational practice requirements, and performance and verification testing requirements for containment zones where human and terrestrial animal pathogens and toxins are handled or stored. By condition of licence or animal pathogen import permit, the Canada Biosafety Standard establishes the criteria for any containment zone where human or terrestrial animal pathogens or toxins are to be safely handled or stored.
The Canadian Biosafety Handbook is a national guidance document for the safe handling and storing of human and terrestrial animal pathogens and toxins in Canada. A companion document to the Canadian Biosafety Standard, the Canadian Biosafety Handbook provides core information and guidance on how to achieve the biosafety and biosecurity requirements specified in the Canadian Biosafety Standard.
The Canadian Biosafety Handbook addresses the development and maintenance of a comprehensive risk-based biosafety management program. In addition to the Standard and Handbook, Canada has national guidelines for containment level 1 physical design and operational practices, developing a biosecurity plan, and for notifying and reporting under the Human Pathogens and Toxins Act and the Human Pathogens and Toxins Regulations.
Canada's national biosafety and biosecurity regulatory oversight under the act and regulations has the following six components:
- Licensing
- Human Pathogens and Toxins Act security clearances
- Exposure reporting and surveillance
- Pathogen risk assessment
- Learning and knowledge
- Compliance verification, monitoring and enforcement
As specified in section 7(1) of the act, unless otherwise exempted, a licence must be obtained from PHAC to authorize any of the following controlled activities with human pathogens and toxins:
- Possessing, handling or using a human pathogen or toxin
- Producing a human pathogen or toxin
- Storing a human pathogen or toxin
- Permitting any person access to a human pathogen or toxin
- Transferring a human pathogen or toxin to another facility
- Importing or exporting a human pathogen or toxin
- Releasing or otherwise abandoning a human pathogen or toxin
- Disposing of a human pathogen or toxin
Public Health Agency of Canada issues licences under the Human Pathogens and Toxins Act to authorize one or more controlled activities with human pathogens and toxins. The licence specifies which of the controlled activities identified in section 7(1) of the act are authorized and the facility or facilities in which the controlled activities are authorized.
The person to whom the licence has been issued is identified as the "licence holder." The act and regulations detail the specific requirements and obligations of licence holders. The general conditions that apply to every licence are specified in section 4 of the regulations. Additional conditions of licence may also be imposed (Human Pathogens and Toxins Act 18[4]). Compliance with the Canadian Biosafety Standard is a condition of licence.
Biological safety officers keep an updated list of pathogens and toxins that are in the possession of the institution. PHAC keeps a national inventory of which risk groups are held within each facility. For dangerous pathogens and select toxins the exact inventory and location are recorded. Moderate risk pathogen and toxin inventories are maintained and kept at the institutions and reviewed upon inspection or request. The licence holder is responsible for ensuring all persons conducting controlled activities are aware of the licence conditions. A biological safety officer is designated by the licence holder to help implement the licence requirements. The role of the biological safety officer is described in the act (sec 36), regulations (sec 9.1) and Canadian Biosafety Standard (sec 4.1).
The importation into Canada of an animal pathogen or part of one that retains its pathogenicity-or animals, animal products, animal by-products, or other organisms that carry an animal pathogen or part of one that retains its pathogenicity-is regulated by PHAC or the CFIA under the Health of Animals Act and Health of Animals Regulations. Importation of animal pathogens under the act and regulations requires an animal pathogen import permit from PHAC or the CFIA. The act and regulations detail the requirements and obligations of a person handling material imported under an animal pathogen import permit.
The CFIA's Office of Biohazard, Containment and Security under the Health of Animals Act and regulations issues certificates to facilities hosting activities on foreign animal disease and emerging animal disease agents. The biocontainment facilities are inspected, audited, assessed and certified to ensure they meet the physical and operational requirements listed in the Canadian Biosafety Standard, 2nd Edition, 2015. The certificate specifies the pathogens and controlled activities and programs that can be carried out in the authorized facilities.
The Plant Protection Act, Seed Act, and the Canadian Containment Standards for Facilities Handling Plant Pests, 1st Edition, 2007, give the CFIA the authority to prohibit or restrict the movement into, within and out of Canada of any plant pest or other thing that is or could be infested with a pest, or is or could be a biological obstacle to the control of a plant pest.
The Canadian Containment Standards describe the physical and operational requirements for facilities that work with plant pests. The importation into Canada of plant pests, plants or plant products and growing media that carry a plant pest is regulated by the CFIA. Importation and domestic movement of plant pests require a plant pest import permit or a domestic pest movement certificate from the CFIA.
Canada's Export Controls Handbook is a reference tool to assist exporters with questions about Canada's export controls, which are administered pursuant to the Export Control List under the authority of the Export and Import Permits Act. The Handbook contains information on how to obtain the necessary permits for the export or transfer of controlled items and how to comply with the requirements of the Export and Import Permits Act.
Federal outreach
PHAC promotes awareness and conducts inspections to monitor compliance. These activities are based on the premise that the majority of the regulated community will comply with legislative and regulatory requirements if they understand the requirements and have the tools to comply. The federal government publishes laws, regulations and guidelines and engages in outreach and education activities, including:
- Direct communication with stakeholders through consultations, email and the Biosecurity Portal
- Compliance training
- Publication and distribution of Canadian Biosafety Standard, Canadian Biosafety Handbook, and Guidelines
- Distribution of Pathogen Safety Data Sheets
- Biosafety advisories and notifications
Multiple federal regulatory and law enforcement departments and agencies participate in Public Safety Canada's Safeguarding Science initiative to educate academia and the private sector about dual use, security and proliferation risks associated with the use of biological and nuclear materials.
Monitoring activities, inspection and enforcement
PHAC's Compliance and Enforcement Policy outlines its graduated approach to compliance and enforcement. It begins with the least intrusive, such as issuance of letters of non-compliance, and moves progressively to more serious interventions, such as revocation of the licence, seizure, detention and disposal. The CFIA may also use administrative monetary penalties as a regulatory enforcement mechanism.
Compliance is normally achieved through a cooperative approach, but in some cases enforcement actions are needed to correct or prevent non-compliance. Transport Canada also uses a graduated approach and has multiple enforcement tools, such as ticketing for non-compliance, detention notices, notices of direction to remedy non-compliance and notice of direction not to import or to return to place of origin.
PHAC has a law enforcement mandate under the Human Pathogens and Toxins Act and, in some cases, investigates and recommends prosecution to the Public Prosecution Service of Canada. The CFIA and Transport Canada conduct penal enforcement activities respectively under the Health of Animals Act, the Plant Protection Act and the Transportation of Dangerous Goods Act.
The biosafety and biosecurity requirements outlined in the Canadian Biosafety Standard are used by PHAC and CFIA when they monitor regulated facilities and importations of animal pathogens or plant pests as part of the application or renewal process for a licence, permit, or facility certification.
Facility certification is the CFIA's formal acknowledgement that a containment zone or facility
complies with the physical containment, operational practice, and performance and verification testing requirements specified in the Canadian Biosafety Standard or the Canadian Containment Standards. Before issuing an animal pathogen import permit, the CFIA must be satisfied that the activities for which the permit is issued would not result in the pathogen's introduction into Canada or into another country from Canada, or its spread within Canada (Health of Animals Regulations 160[1.1]). Applicants of an animal pathogen or plant pest import permit may be subject to facility certification or compliance verification to demonstrate that the containment zone meets requirements.
For higher risk containment zones, the facility certification process may include an onsite inspection and a review of as-built drawings with specifications. During the commissioning of the facility, it may also include performance and verification testing of critical containment components, the Biosafety Manual, the containment zone standard operating procedures, and, for work with RG4 pathogens, a review of training records.
Facilities certified by the CFIA may require annual recertification to verify ongoing compliance, including a review of documentation, such as program intent and the performance and verification testing reports of critical containment systems.
Inspections of laboratories are an important compliance monitoring activity. PHAC's risk-based approach to selecting which laboratories to inspect and how frequently takes into consideration risk factors, such as compliance history, quality of the facility's biosafety program, category of pathogen (with RG4 inspections every year and RG3 inspections every 3 years), and the complexity of the work done at the facility.
Human Pathogens and Toxins Act and Health of Animals Act inspectors and each facility's biological safety officer are responsible for monitoring compliance at licenced facilities. The function and authority of inspectors is described in the Human Pathogens and Toxins Act sections 40 to 52 and in the CFIA Act subsection 13(3). Biological safety officers have the appropriate knowledge for the containment levels and pathogens and toxins handled at their facility and they support:
- Accuracy and completeness of all licence, animal import and material transfer applications
- Communication with PHAC and the CFIA
- Promotion and monitoring of facility's compliance with legislation, regulations, and standards
- Development and maintenance of biosafety manual and standard operating procedures
- Quality of internal incident investigations
Global Affairs Canada's Trade Controls Bureau is responsible for administering the Export and Import Permits Act. The Canada Border Services Agency and the Royal Canadian Mounted Police enforce it. Where offences are suspected, border services officers may detain or seize goods and forfeiture action may be taken. Corporations and their officers who contravene the Export and Import Permits Act are potentially liable. Investigations can lead to charges, prosecutions, fines and/or incarceration.
The Guidelines for the Notification and Testing of New Substances help individuals responsible for complying with the New Substances Notification Regulations (Organisms) under the Canadian Environmental Protection Act 1999 comply with the rules. Enforcement of New Substances Notification Regulations (Organisms) is the responsibility of Environment and Climate Change Canada enforcement branch.
The Customs Act enables the Canada Border Services Agency to ensure that goods exported from Canada comply with export controls under all acts of Parliament. Border services officers perform compliance monitoring and enforcement at the border.
Laboratory accreditation for biosafety and biosecurity
Although external accreditation is not required under the Human Pathogens and Toxins Act and the Human Pathogens and Toxins Regulations, many laboratories in Canada seek accreditation to comply with other registration bodies or clients. Accreditation can be done through organizations such as provincial colleges of physicians and surgeons, the College of American Pathologists and Accreditation Canada or through compliance with International Organization for Standardization standards.
The accreditation process typically includes rigorous on-site inspections in which auditors examine records, staff qualifications, equipment, facilities, safety records and overall management of the laboratory. Although these processes are primarily focussed on diagnostic quality, there is some overlap with biosafety and biosecurity requirements (see Laboratory audits subsection below).
Funding
As federal programs, all the components of Canada's biosafety and biosecurity activity receive stable and adequate funding through the federal government's annual budgeting process. PHAC reviews its biosafety and biosecurity oversight program every five years to assess value, effectiveness and reach.
Oversight of dual use research
Regulated parties who intend to carry out scientific research under the Human Pathogens and Toxins Regulations must submit, as part of their Human Pathogens and Toxins Act licence application, a plan for administrative oversight of pathogens and toxins in a research setting. The plan eliminates gaps in the oversight of pathogens at the institutional level by establishing effective accountability structures.
Institutions must also ensure that their scientists are aware of potential dual use risks. The Canadian Biosafety Standard (section 2.3.5) describes how to identify dual use risks and PHAC supports awareness through guidelines and training, such as the online Introduction to Dual Use in Live Science Research that encourages responsible conduct of research among scientists, administrators, funding organizations, policy-makers, and the public.
Physical security
All Human Pathogens and Toxins Act licence holders in Canada must have security measures in place to minimize inappropriate removal or release of biological agents. They must also meet physical containment requirements in the engineering and design of their facility. The Canadian Biosafety Standard also requires all Human Pathogens and Toxins Act licence holders and Health of Animals Act permit holders to conduct a biosecurity risk assessment (Canadian Biosafety Standard section 4.1.7) and to develop, implement and evaluate a biosecurity plan to address physical security (Canadian Biosafety Standard section 4.1.11).
Provincial and territorial facilities must participate in the national licencing program. Their governments help licence holders meet those national requirements. For example, Alberta publishes Best Practices for the Assessment and Control of Biological Hazards; Saskatchewan has the Medical Laboratory Quality Assurance Program administered by the College of Physicians and Surgeons to support compliance; and British Columbia is currently developing a comprehensive biosecurity plan for the province. In Quebec, biomedical laboratories must meet the requirements under section 7.1 of the Qmentum Program.
Information security
In Canada access to sensitive information is controlled by a variety of policies and procedures. At the federal level these include:
- Archived: Policy on Government Security, which provides direction on government security to support program and service delivery, and to protect information and individual assets
- Archived: Directive on Departmental Security Management, which supports departments in achieving efficient, effective and accountable management of security
- Archived: Security Organization and Administration Standard, which establishes the standard required by the Security policy (above) and has sections on information and information technology security
- Archived: Operational Security: Management of Information Technology Security Standard, which defines baseline security requirements for federal departments to ensure the security of information and information technology assets under their control.
The Canadian Biosafety Standard includes requirements for restricting access to sensitive information.
Provinces and territories have additional legislation, policies and procedures in place to control access to sensitive information. For example, Alberta's Health Information Act controls access to sensitive health information and Quebec follows standard 24.6 of the Qmentum program addressing information security measures. Finally, the biosecurity risk assessment and plan, which all regulated facilities in Canada must have in place as a condition of their licence, addresses information security (Canadian Biosafety Standard section 4.1.11).
Transportation safety
Transport Canada administers the Transportation of Dangerous Goods Act and Archived: Transportation of Dangerous Goods Regulations, which control the way toxic and infectious substances, including waste, can be imported, transported or shipped. Each province and territory in Canada has adopted the regulations as its own legislation. The act identifies 9 classes of dangerous goods, including toxic and infectious substances (class 6.1 and 6.2 respectively). The regulations include a list of class 6.2 substances by category and describe the shipping requirements for these substances in detail-from containment and labeling to documentation and training.
The Transportation of Dangerous Goods Act (Part 7) requires that before a person handles, offers for transport, transports or imports certain classes of dangerous goods, they must have an approved Emergency Response Assistance Plan in place that describes what is to be done in the event of an accident. Transport Canada verifies and approves these plans under the Act. The National Microbiology Laboratory is the national coordinator for the Emergency Response Assistance Plan program to deal with national emergencies involving human pathogens.
Personnel security and professional qualifications
All licenced facilities in Canada do a biosecurity risk assessment and develop a plan that addresses personnel suitability and reliability. Consequently, they all have procedures for assessing an individual's suitability and reliability. In addition, an HPTA security clearance is required for people who have access to Security Sensitive Biological Agents toxins and pathogens that pose a risk to Canada's national security if misused. The HPTA Security Clearance Program validates that people who have access to Security Sensitive Biological Agents are reliable and trustworthy and do not pose a security risk by virtue of their access.
PHAC provides resources on its e-learning portal to help regulated parties complete their personnel training requirements. For more information on training see the section on Training at laboratory facilities below.
Facility emergency planning
Human Pathogens and Toxins Act licenced facilities have site-specific biosafety and biosecurity management plans in place, such as a biosafety manual, biosecurity plan and standard operating procedures. These materials are informed by risk assessments mandated under the Canadian Biosafety Standard and biological safety officers help develop and maintain them.
Any facility that intends to carry out scientific research must also have a plan in place that sets out administrative measures for managing and controlling biosafety and biosecurity risks in a research setting. The Canadian Biosafety Standard outlines requirements for incidence and emergency response and reporting (Canadian Biosafety Standard 4.9), as well as administrative requirements for records and documentation (Canadian Biosafety Standard 4.10).
Examples include the National Microbiology Laboratory's Incident Response Manual and Biosafety Manual; Alberta Health Services' Code Brown Hazardous Spills Plan and Best Practices for the Assessment and Control of Biological Hazards; and the Northwest Territories' Stanton Territorial Health Authority Biosafety Program Manual. Manitoba's Safety and Risk Management Policy outlines roles and responsibilities in the province related to biosafety and biosecurity management.
Laboratory audits
Laboratories in Canada are audited and/or inspected by PHAC (as national regulator) and/or the CFIA, the facility's biological safety officer, provincial occupational health and safety bodies, and accredited facilities are also audited by external accreditation bodies. Human Pathogens and Toxins Act licenced facilities are required to perform internal inspections and audits at a frequency determined by a local risk assessment.
Some provinces and territories have accreditation or licencing programs for medical/diagnostic laboratories. There are minor differences in the audit systems among provinces and territories. For example, British Columbia has a Mandatory Provincial Laboratory Accreditation Program and Diagnostic Accreditation Program and the provincial laboratory is also accredited through the College of American Pathologists Program. BC's CL2 and CL3 laboratories also have their own internal quality assurance processes.
In Manitoba internal and external safety audits of all laboratory areas are conducted quarterly. Some facilities are audited by external accrediting bodies, while internal audits are conducted by the Workplace Health and Safety Committee or the safety and compliance officer. Manitoba's public health focused laboratory is accredited by the College of American Pathologists. Laboratories in the Northwest Territories are inspected on a daily, monthly and annual basis by hospital staff, following the government's Stanton Territorial Health Authority Biosafety Program Manual.
External audits are conducted by various levels of government or arm's length agencies, for example:
- The laboratory at Stanton Territorial Hospital (Northwest Territories) is accredited through Accreditation Canada.
- Nova Scotia has a program of annual biosafety and biosecurity audits conducted by the provincial laboratory's biological safety officer.
- Laboratories in Quebec (Available in French only) and Prince Edward Island's laboratories are accredited through Accreditation Canada to ISO 15189.
- In Saskatchewan the provincial standards are based on the ISO standard.
- In Ontario, under the Laboratory and Specimen Collection Centre Licensing Act, all clinical laboratories and specimen collection centres must meet the Institute for Quality Management in Healthcare requirements (based mainly on ISO 15189) as a condition of licence. The Institute does assessments as part of accreditation. However, the Ministry of Health and Long-Term Care also has the power to inspect laboratories for compliance with the Laboratory and Specimen Collection Centre Licensing Act.
The National Microbiology Laboratory has accreditation in ISO 9001, ISO 15189, ISO 17025, and ISO 17043 and undergoes internal biosafety and biosecurity audits and external ISO quality audits. Biological safety officers and bio-risk officers do internal audits following the Biological Inventory Assessment Protocol.
The scope of these internal audits may also be assessed by PHAC and the CFIA when they inspect facilities under the HPTA and HAA. Quality, system-based internal audits are performed by trained Quality Office and laboratory personnel as per internal audit procedures.
National laboratories and relevant classifications
PHAC is a designated World Health Organization (WHO) Collaborating Centre for biosafety and biosecurity, while the National Microbiology Laboratory serves as:
- WHO Regional Reference Centre for Measles and Rubella
- WHO National Influenza Centre for Canada
- WHO Collaborating Centre for Emerging and Zoonotic Diseases Detection, Diagnostics, Reference and Research
- PAHO/WHO Polio Regional Reference Laboratory
The CFIA is the Canadian delegate of the World Organisation for Animal Health and its laboratories serve as the World Organisation for Animal Health Collaborating Centre for Food-Borne Zoonotic Parasites and as the Organization's Reference Laboratory for Rabies, Scrapie, Chronic Wasting Disease, Avian Influenza, Classical Swine Fever and Trichinellosis.
Through a number of international partnerships and networks-such as the European Commission, the Global Health Security Action Group Laboratory Network, and the Biosafety Level 4 Zoonotic Network-the National Microbiology Laboratory has established relationships for sharing information and best practices to respond to threats of emerging disease and to monitor global activities.
Reducing access to dangerous pathogens and toxins
The Human Pathogens and Toxins Act and the Human Pathogens and Toxins Regulations do not require dangerous pathogens and toxins to be consolidated in a minimum number of facilities in Canada. Nonetheless, some dangerous pathogens have been consolidated to a minimum number of facilities, for example all RG4 pathogens are consolidated in one jointly operated federal facility in Canada and efforts are underway to reduce the number of facilities in Canada that contain polio virus.
In addition, many Canadian laboratories use diagnostic tests that eliminate the need to culture dangerous pathogens. The National Microbiology Laboratory minimizes risks as much as possible when developing or selecting diagnostic methodologies and many of its programs have shifted to molecular technologies. However, confirmatory assays are still culture-based.
P.6.2 Biosafety and biosecurity training or plans are in place
Training at laboratory facilities
There are training programs in place at all Human Pathogens and Toxins Act licenced facilities in Canada. Biological safety officers are responsible for arranging appropriate training on applicable biosafety and biosecurity laws, regulations and standards at their facilities. Furthermore, the Canadian Biosafety Standard (section 4.3) outlines training program requirements, which are applicable to the Human Pathogens and Toxins Act, the Human Pathogens and Toxins Regulations, the Health of Animals Act and the Health of Animals Regulations.
PHAC provides resources to help laboratories meet training requirements. The Laboratory Biosafety and Biosecurity e-learning portal gives licence holders and stakeholders access to common course material and instructional videos. Although not mandatory, the training material supports a common curriculum for biosafety and biosecurity in Canada.
The current training requirements and course materials are based on a needs assessment conducted in 2012. The Agency, an accredited American National Standards Institute and International Association for Continuing Education and Training biosafety training provider, is working to align its training modules with the American National Standards Institute's International Association for Continuing Education and Training Standard, which is already reflected in the Canadian Biosafety Standard (sec 4.3.9-10).
The National Microbiology Laboratory provides mandatory biosafety and biosecurity training or refresher training based on a common curriculum at all its sites. It maintains a database of staff training results and conducts annual emergency drills to test staff on procedures.
The Transportation of Dangerous Goods Regulations require that any person who handles, offers for transport or transports dangerous goods be adequately trained and hold a training certificate. With few exemptions, carriers who ship, store or handle infectious substances in Canada have appropriate training. Transport Canada refers carriers who require training to private sector companies that provide appropriate training on the Transportation of Dangerous Goods Regulations. The Department is currently standardizing training requirements and updating them to better reflect current industry practices.
Provinces and territories employ a variety of strategies to meet the staff training requirements of their licences. For example, mandatory biosafety and biosecurity training is part of new employee orientation in Manitoba; Alberta has a provincial competency and acknowledgement program; and the Northwest Territories does annual employee training needs assessments. In Ontario, each laboratory and specimen collection centre must provide Institute for Quality Management in Healthcare induction and refresher training on biosafety and biosecurity.
Oversight of the training and exercise requirement
Public Health Agency of Canada and CFIA inspectors review training documentation to determine whether or not a facility is meeting its training requirements and make recommendations for corrective action, if necessary. At the National Microbiology Laboratory, drills and exercises are assessed through a "hotwash" process that includes gathering suggestions for improvements and drafting an action plan to implement corrections.
Funding for training
PHAC's Biosafety and Biosecurity Training Program has adequate, stable funding for the current task of creating and distributing national bilingual biosafety and biosecurity training resources online. Each licenced facility is responsible for finding appropriate internal training programs that allow them to meet their particular licence requirements.
Context: Facilities and biosafety equipment maintenance
Maintenance planning for new facilities
As part of the Human Pathogens and Toxins Act licencing and the Health of Animals Act permit process in Canada, all new facilities where people work with dangerous pathogens are planned with long-term resource commitments, and all high-risk facilities are formally commissioned before opening.
Availability of medical maintenance and waste management
Under the Canadian Biosafety Standard (section 4.8), decontamination and waste management procedures must be documented in standard operating procedures that are easily understood and implemented by all personnel. The Canadian Biosafety Standard (section 5.1.5) also requires that class II biological safety cabinets be certified in accordance with National Sanitation Foundation International and American National Standards Institute 49 Biosafety cabinetry design, construction, performance, and field certification.
Licenced facilities in provinces and territories have appropriate waste management policies in place and well-documented procedures. For example, Alberta's Waste Management Policy and IPC Community-Based Services Resources Manual: Waste and Sharps Handling; and the Northwest Territories' Stanton Territorial Health Authority Biosafety Program Manual (Appendix 36). In all provinces and territories, except Prince Edward Island and the Northwest Territories, biosafety cabinets can be serviced locally. HEPA Atlantic, located in Nova Scotia, certifies and maintains Prince Edward Island's biological safety cabinets and the Northwest Territories has a training program in place to build local capacity.
Human Pathogens and Toxins Act licence holders and Health of Animals Act permit holders are responsible for maintaining their facilities as specified in the provisions of the Canadian Biosafety Standard. Funding for facility and equipment maintenance varies from province to province-most have adequate funding set aside for timely maintenance but some facilities report that they need additional funding to keep equipment current (Manitoba and Northwest Territories).
Transportation of biological material
Canada's Transportation of Dangerous Goods Regulations covers category A and B infectious substances and is based on the United Nations' Recommendations on the Transport of Dangerous Goods Model Regulations. The Transportation of Dangerous Goods Regulations is updated regularly to harmonize Canadian regulations with UN recommendations. Transport Canada and its provincial and territorial partners enforce the regulations as set out in existing memoranda of agreement between the federal government and each province and territory. The regulations apply to dangerous goods being imported, handled, offered for transport, or transported by air, marine, rail and road modes of transportation.
The Canadian Biosafety Standard sets out requirements for movement and transport of pathogens and toxins within laboratories, and between laboratories located inside the same building as these movements are not included in the scope of the Transportation of Dangerous Goods Regulations (Canadian Biosafety Standard requirements 4.6.21 and 4.8.8).
In provinces and territories, carriers must follow both the national regulations and any relevant provincial or territorial regulations, such as Alberta's Dangerous Goods Transportation and Handling Regulation and Transportation of Infectious Substances guide.
Occupational health and safety
Workplace safety, including safety in laboratories, is generally a matter of federal, provincial and territorial jurisdiction. Provinces and territories are responsible for occupational health and safety and have employer Workplace Hazardous Materials Information System (WHMIS) requirements within their respective jurisdictions. Provincial and territorial occupational health and safety agencies appoint inspectors under their legislation to verify compliance and take measures to confirm work is being carried out safely.
Laboratory personnel in federal and most provincial and territorial laboratories have equal access to occupational health and safety services. Personal protective equipment is readily available in all facilities and immunization policies and standards for laboratory workers (as health care workers) are in place in most provinces and territories. For example:
- Alberta Health Services has an immunization policy and standard for health care workers.
- New Brunswick has a vaccination policy for laboratory personnel.
- Northwest Territories Stanton Territorial Health Authority Biosafety Program Manual covers vaccinations for laboratory personnel.
- Quebec has a guide (Available in French only).
- Newfoundland and Labrador has a provincial immunization policy manual.
- Prince Edward Island has the Recommended Immunization Schedule for Health Care Workers.
In Ontario, the Occupational Health and Safety Act and the accreditation program for laboratory and specimen collection centres promote occupational health services.
Post-exposure prophylaxis is provided to health care workers, including laboratory workers, in facilities across Canada. Most laboratories have post-exposure prophylaxis guidelines, protocols, or plans, such as:
- Alberta's guidelines for non-occupational, occupational and Mandatory Testing and Disclosure Act post exposure management and prophylaxis
- Prince Edward Island's Guidelines for the management of a percutaneous or sexual exposure to blood borne Pathogens
- Quebec has developed the Guide pour la prophylaxie et le suivi après une exposition au VIH, au VHB et au VHC (Available in French only)
The Human Pathogens and Toxins Act (sections 12,13 and 14) and the Canadian Biosafety Standard (section 4.1) set out notification and reporting requirements for licence holders, biological safety officers, and persons conducting controlled activities authorized under a licence. To facilitate notification to PHAC, an on-line Biosecurity Portal (LINC) is available for secure creation, storage and submission of laboratory incident reports and other notifications. Mandatory notification to the Agency is required without delay when an incident involving a human pathogen or toxin has caused, or may have caused, disease in an individual.
Best practices, challenges, gaps and recommendations
Canada's current biosafety and biosecurity oversight framework is very comprehensive and risk based. It covers all sectors working with risk group 2, 3 and 4 pathogens and selected toxins, and its controls are commensurate with the risks. A performance based approach provides multiple means to achieve the same objective, which is the safe and secure use of pathogens and toxins. Increased institutional accountability fosters a culture of risk management within regulated institutions.
From a federal perspective, it is also continually improved through the "life cycle" approach to regulation making articulated in the Cabinet Directive on Regulatory Management. The life cycle approach recognizes that attention must be given not only to regulatory development and analysis but also to the implementation, evaluation, and review of regulations. The life cycle approach improves the effectiveness, efficiency, and accountability of the regulatory system to support the government's commitment to Canadians. A further consultative approach is used to promote policy interventions that are sustainable within the regulated community.
The Government of Canada recognizes that the oversight framework could be enhanced. Canada aims to strengthen biosafety and biosecurity practices through specific deliverables including:
- Optimizing delivery of the biosecurity program
- Addressing potential regulatory duplication among federal authorities
- Recommending action for collaboration between federal partners
- Exploring innovative means to further educate regulated parties
Canada recognizes the challenge of emerging technologies and the need for an oversight framework on dual use in life sciences research and is currently increasing coordination with stakeholders in the other federal departments, provincial and territorial governments, industry, education, research, and the public in order to strengthen an integrated oversight framework that raises awareness of the impact of dual use research at various steps within the research continuum.
P7: Immunization
Joint external evaluation target: A functioning national vaccine delivery system-with nationwide reach, effective distributions, access for marginalized populations, adequate cold chain, and ongoing quality control-that is able to respond to new disease threats.
Level of capability in Canada
In Canada, vaccination is a responsibility shared among the federal, provincial and territorial governments. Provinces and territories deliver the vaccination program, which includes selecting and funding vaccines for their populations, determining their vaccination schedules, designing and delivering immunization programs and monitoring vaccine uptake within their populations. It is important to note that vaccines are not covered by the Canada Health Act and are therefore provided as supplementary services at the discretion of each province and territory.
The federal government has a role in regulatory approval, vaccine safety, bulk purchasing of vaccines, national surveillance for vaccine-preventable diseases and adverse events following vaccination. It also assumes responsibility for national leadership and coordination in knowledge development, innovation, and sharing best practices.
The National Immunization Strategy, established in 2003 by federal, provincial and territorial Deputy Ministers of Health, provides a framework for effective inter-jurisdictional collaboration that improves the relevance, effectiveness, and efficiency of immunization programing across Canada. At the time of its creation, the National Immunization Strategy was designed to address a number of immunization challenges in Canada.
Work under the National Immunization Strategy by all jurisdictions continues through national engagement and collaboration under the Public Health Network Council structure, particularly through the Canadian Immunization Committee and the Vaccine Supply Working Group. The result is that jurisdictions today are better connected, helping ensure that immunization program design and delivery is evidence-based and sustained by a safe and secure vaccine supply chain, which benefits all Canadians.
Canada's updated National Immunization Strategy Objectives 2016-2021 aligns with the World Health Organization (WHO) Global Vaccine Action Plan's principles (ownership, partnership, equity, integration, sustainability and innovation) and strategic objectives.
Indicators
P.7.1 Vaccine coverage (measles) as part of national program.
Current Canadian programs supporting vaccination (in general)
In Canada, responsibility for health care, including immunization, is shared by the federal, provincial and territorial governments. While each jurisdiction has a distinct mandate and unique operating context, their activities are complementary and collaborative.
Vaccine authorization
Health Canada conducts rigorous scientific review and testing of vaccines to assess their quality, safety and efficacy before they are approved for use in Canada. Once a vaccine is licenced for use in Canada, vaccine safety is monitored to detect and respond to adverse events following immunization. Under the Food and Drugs Act and regulations, vaccine manufacturers are required to report to Health Canada any serious adverse events of which they become aware. Most provinces also require vaccination providers to report serious adverse events to provincial public health authorities.
Vaccine recommendations
Recommendations on the use of vaccines in Canada are provided by the Public Health Agency of Canada, and developed through its external advisory body, the National Advisory Committee on Immunization. This group of experts provides transparent technical and scientific analysis and recommendations on new and existing vaccines. Provinces and territories use these vaccination recommendations to assist in the planning of their vaccination programs.
The Committee to Advise on Tropical Medicine and Travel is an expert advisory body that assists the Public Health Agency of Canada with travel health-related advice for travellers and health care professionals. Country-specific vaccine recommendations, along with other related resources, are publicly available on the Government of Canada's travel health website.
Vaccine procurement
The federal government aims to enhance affordability of the vaccine supply in Canada by facilitating provincial and territorial participation in the Bulk Procurement Program to benefit from multi-year contracts with multiple providers. Under the Program, smaller jurisdictions can purchase vaccines at a lower cost than if purchased independently.
Vaccination programs
Canadian provinces and territories implement vaccination programs that align with the goals of their respective health care plans. They take into consideration recommendations, scientific evidence, local epidemiology of diseases, budgets, and public health capacity. They all have programs that provide publicly-funded, universal access to vaccines for infants, children and adolescents. These include vaccines against diphtheria, pertussis, tetanus, polio, H. influenzae type B, hepatitis B, measles, mumps, rubella, varicella, invasive meningococcal disease, invasive pneumococcal disease, and human papillomavirus.
Publicly-funded adult vaccination programs provide universal access to vaccines for tetanus and diphtheria (Td), influenza for pregnant women, adults with chronic medical conditions, and adults 60 or 65 years and older, as well as pneumococcal polysaccharide vaccine for seniors aged 65 years and older.
Indicators
National vaccination coverage goals were developed for childhood, adolescent and adult vaccines that are publicly funded in all provinces and territories. The goals are based on best practices, international standards, and a review of existing data and surveillance systems to set baseline metrics. These include a vaccination goal of 95% coverage for childhood vaccines, 90% coverage for adolescent vaccines, and specific goals for older adults and healthcare workers for relevant vaccines.
Monitoring national vaccination coverage (in general)
The Public Health Agency of Canada conducts national population surveys to report on national vaccine uptake for children and adults, and for seasonal influenza. To gain a clearer understanding of the factors influencing decisions on immunization, the surveys include questions on parental knowledge, attitudes and beliefs, as well as vaccine hesitancy and barriers to immunization.
National immunization coverage estimates provide information on the level of protection against disease in Canada and identify general trends over time in uptake and attitudes toward vaccines. Survey results are used to measure progress towards Canada's national vaccination goals and to provide estimates of coverage to the World Health Organization and the Pan American Health Organization.
In 2015, Canada's vaccination coverage estimates by two years of age were:
- 89% for measles (one dose)
- 91% for diphtheria, pertussis, tetanus and polio (3 doses)
Provinces and territories monitor vaccine coverage using data from different sources, including provincial immunization registries or repositories (if available), day care or school entry records, or results of provincial vaccination coverage surveys (table 3).
Systems for identifying and addressing disparities
Analysis of the 2013 Childhood National Immunization Coverage Survey indicated some inequalities in vaccine uptake. Children of low socio-economic status were at higher risk of being never vaccinated or having incomplete vaccination. The results also indicate a strong association between parents' lack of confidence in vaccine effectiveness and safety and incomplete immunization. In contrast, analysis of data related to human papillomavirus vaccination indicates that higher parental education level has been associated with refusal of the vaccine.
The socio-demographic data and sample size of the national immunization coverage surveys only allow for limited analysis of the determinants of non-vaccination, under-vaccination and barriers to vaccination. National coverage assessment methods did not:
- Identify under and non-vaccinated sub-population groups at the community level
- Describe the extent of under and non-vaccination among various sub-populations, including some potentially vulnerable sub-populations, such as recent newcomers to Canada, Indigenous peoples
- Determine the systemic barriers negatively impacting vaccination access and uptake
Initiatives to address these identified gaps are being developed and implemented under the Government of Archived: Canada's 2016 Budget, in which the Government of Canada committed "to improve Canada's ability to identify under- and un-immunized Canadians and develop a focused program to improve vaccine access and uptake," and through the National Immunization Strategy Objectives 2016-2021 (described in Best practices, challenges, gaps and recommendations).
At the local level, data from immunization registries and school programs have been used to identify neighbourhood immunization rates and disparities. For example, this approach was successfully implemented by the Saskatoon Health Region to identify disparities and reallocate resources to improve vaccine coverage for the entire population.Footnote 3
P.7.2 National vaccine access and delivery
Appropriate management of vaccine stock
The federal government through Public Services and Procurement Canada operates a bulk procurement program for vaccines. The Public Services and Procurement Canada program, which contracts with multiple suppliers for a given vaccine, allows Canada to manage vaccine shortages quickly and ensure equitable, affordable access for all jurisdictions. Approximately 85% of vaccines for publicly funded vaccination programs in Canada are purchased through the program. Provinces and territories that decide to join the program determine their own requirements and pay for the vaccines they use.
The Public Health Agency of Canada co-chairs the Canadian Immunization Committee's Vaccine Supply Working Group, a multi-jurisdictional group that monitors vaccine supply and prices and develops principles, strategies and guidelines to address vaccine supply issues (shortages, quality) and to facilitate fair and equitable distribution of vaccines during a shortage.
In addition to the federal government mechanisms to ensure sustainable supplies of vaccines (mentioned above), provinces and territories rely on best practices, such as good forecasting, inventory monitoring, central oversight, and safety stocks at provincial depots to maintain supply of vaccines.
Vaccine warehouses in Canada must have a Drug Establishment Licence and Good Manufacturing Practice certification from Health Canada. Federal inspectors ensure that warehouses maintain standards for cold chain storage and distribution. Vaccine contracts under the bulk procurement program for vaccines describe the requirements for cold chain maintenance. The supplier maintains the cold chain during delivery to provincial or territorial depots across the country.
Once the vaccine is accepted at a depot, the province or territory is responsible for maintenance of the cold chain during storage and further distribution of the vaccine. The Public Health Agency of Canada issued Archived: National Vaccine Storage and Handling Guidelines for Immunization Providers in 2015. Provinces and territories have policies and issue guidance on cold chain management for public health and health professionals involved in vaccine delivery.
Distribution of vaccine
Although vaccination is a personal choice in Canada, it is strongly encouraged by federal, provincial and territorial authorities. The federal government supports education and promotion campaigns to encourage vaccination, while provinces and territories use a variety of strategies from education and vaccine awareness campaigns to regulations that require reporting vaccination status for school or day care entry.
Vaccination delivery and information systems in Canada
Most provinces and territories offer information about vaccines on their websites and make vaccines available through a network of health care providers (public health nurses, pharmacists, physicians) in a variety of settings. Vaccination delivery models vary across Canada (Figure 4). British Columbia, Manitoba, Ontario, Quebec, New Brunswick, Nunavut, and Nova Scotia have mixed models involving primary health care providers and public health clinics. Prince Edward Island, Newfoundland and Labrador, Saskatchewan, Alberta, the Northwest Territories and Yukon Territory rely mainly on public health for vaccination delivery.
All provinces and territories deliver school-based vaccination programs (where nurses vaccinate students in temporary clinics set up in schools) for vaccines recommended for school-age children and youth (i.e. hepatitis B vaccine and the human papilloma virus vaccine). This approach optimizes access to vaccines for children and youth.
Pharmacists' scope of practice has expanded in recent years, with vaccine injection authority in all provinces and territories except Quebec, Yukon, Nunavut, and the Northwest Territories. The minimum age for clients to be eligible for pharmacist administered vaccine varies by jurisdiction. Pharmacists play an important role in the delivery of the seasonal influenza vaccine, accounting for >50% of doses given in some jurisdictions.
Depending on the vaccination delivery system, received vaccinations are recorded in the child's medical record, parent-held record, administrative data, school board record and, when available, in a provincial or territorial immunization information system. The type of vaccination delivery program influences the completeness of information found in these records.

Figure 4: Immunization delivery and information systems in Canada - Text description
This figure uses a map of Canada to describe the immunization registry landscape in Canada and looks at both immunization delivery and information systems used across the country. Vaccination delivery models vary across Canada. The map indicates that British Columbia, Manitoba, Ontario, Quebec, New Brunswick, Nunavut, and Nova Scotia have mixed models involving primary health care providers and public health clinics. The map highlights that Prince Edward Island, Newfoundland and Labrador, Saskatchewan, Alberta, the Northwest Territories and Yukon Territory rely mainly on public health for vaccination delivery.
The majority of provinces and territories (12 of 13) are in the process of implementing electronic immunization registries. The map indicates that Yukon Territory, British Columbia, Saskatchewan, Manitoba, Ontario, Quebec and Nova Scotia all use Panorama. The remaining jurisdictions use other registries or repositories. The map shows that: Alberta uses Imm/ARI; Northwest Territories uses a Manual database; Newfoundland and Labrador uses CRMS; New Brunswick uses CSDS+3; PEI uses ISM and Nunavut does not employ an electronic registry or repository at this time.
Immunization registries have the capacity to quickly and accurately capture data on the administration of vaccine doses at the individual level and are an important tool in determining the level of protection in a population against vaccine-preventable diseases. The majority of provinces and territories (12 of 13) are in the process of implementing electronic immunization registries, however these are not yet fully functional and do not have the ability to report coverage rates on the entire population.
Provinces and territories are using various strategies to improve the completeness of records in their immunization registries. These include:
- Providing web portal access to the public and health professionals
- Working with electronic medical record vendors to enhance interoperability between electronic medical records and registries
- Enacting legislation to make it mandatory for health care professionals to enter immunizations directly into the registry system
Immunization programs led by the Government of Canada
First Nations living on reserve
The federal government provides primary care and public health services to First Nations living on reserve through policies to improve access to services. This includes providing for immunization services and delivering provincially purchased vaccines according to provincial schedules. Health Canada works with the local public health unit to ensure cold chain integrity during the transportation of vaccines from provincial depots to First Nations communities through the application of provincial standards and guidelines.
Health Canada either directly delivers, or financially supports First Nations communities and/or organizations in the delivery of culturally appropriate immunization programming in First Nations on-reserve communities in Alberta, Saskatchewan, Manitoba, Ontario, Quebec and the four Atlantic provinces. Health Canada's immunization programs work to ensure that immunization services on-reserve align with programming delivered by provincial governments to the general population. It produces culturally relevant education and awareness products targeted to First Nations on-reserve and supports education and awareness activities for Inuit populations in collaboration with its national Inuit partner, Inuit Tapiriit Kanatami.
Health Canada also monitors immunization coverage among First Nations living on reserve to measure progress against national vaccination goals, as well as to identify disparities and to inform program interventions.
Vaccinations are reported through aggregate community-based reporting on an annual basis. Coverage information is further summarized at the regional and national levels. Overall coverage estimates are produced for vaccines where there is limited variation between provincial vaccine schedules. First Nations living on reserve may visit off reserve primary care services for vaccinations. Vaccines received off reserve may not be recorded in community records, which may lead to an under-estimation of coverage estimates for First Nations who live on reserve.
In British Columbia, the First Nations Health Authority has responsibility for health service design and delivery, including immunization programming for First Nations living on reserve. First Nations populations living off reserve and Inuit receive immunization services from the province and territory and Inuit regions in which they reside
Canadian Forces
The Canadian Armed Forces maintains its own Immunization Standard, based on existing national guidance, under which it provides routine vaccinations to service members as well as travel or operationally indicated vaccines (such as rabies, typhoid, yellow fever). The Archived: Canadian Forces Health Information System is used to electronically record all everyday management and delivery of health and dental services, including immunizations.
The Directorate Force Health Protection also collects statistics from individual Canadian Forces clinics on influenza vaccination rates (and specifically immunization of uniformed health care and dental workers). The Department of National Defense participates in Canada's Vaccine Supply Working Group (described previously). In terms of program delivery, Canadian Forces Bases are staffed with dedicated immunization nurses, who deliver vaccination services.
Immigration, Refugees and Citizenship Canada
The Government of Canada's Interim Federal Health Program provides limited, temporary coverage of health-care benefits in Canada to resettled refugees, asylum seekers, and certain other groups, until they become eligible for provincial or territorial health-care coverage. The Program provides basic coverage (i.e. hospital, physician) similar to provincial or territorial health-care insurance, as well as supplemental services (i.e. vision, urgent dental, counselling) and prescription drugs, including vaccinations, similar to provincial vaccine schedules.
Starting April 1, 2017, the Interim Federal Health Program was expanded to cover certain services for individuals who have been identified for resettlement before they come to Canada. These pre-departure medical services include coverage of certain pre-departure vaccinations aligned with Canadian guidelines. These vaccinations are not mandatory for immigration to Canada; they are only given to individuals who consent.
As well, pre-departure medical services include services to manage communicable disease outbreaks in refugee camps, such as measles or influenza. It is expected that these services will prevent the introduction and spread of vaccine-preventable diseases into Canada, and reduce morbidity and mortality among refugees. In addition, the Program will help maintain measles, rubella and congenital rubella syndrome elimination status in the WHO Americas region; support the WHO Global Polio Eradication Initiative; and facilitate rapid school entry for displaced children.
Ensuring the capability for rapid distribution in an emergency
Canada's pandemic vaccine strategy aims to:
- Provide a safe and effective vaccine for all Canadians as quickly as possible
- Allocate, distribute, and administer vaccine as efficiently, equitably and effectively as possible
- Monitor the safety and effectiveness of pandemic vaccine
In addition to its usual role providing regulatory authorization for vaccines, the federal government also negotiates and establishes ongoing contracts for pandemic vaccine. In the event of a pandemic, Canada would leverage its existing vaccine distribution networks as it did during the H1N1 pandemic, 2009-2010.
Health Canada works to support on-reserve First Nations communities in building, strengthening and testing communicable disease emergency plans that include pandemic influenza response. Health Canada also works with the Public Health Agency of Canada and other federal partners, national and regional First Nations organizations, and provinces to ensure comprehensive and coordinated communicable disease emergency planning and response activities in on-reserve First Nations communities.
Best practices, challenges, gaps and recommendations
While immunization coverage in Canada today is good and progress is being made toward common objectives, more is needed in Canada to reach national vaccination coverage goals. Furthermore, while rates of vaccine-preventable diseases in Canada are low, recent measles and pertussis outbreaks demonstrate that Canadians are still at risk.
In its 2016 Budget, the Government of Canada committed $25M over five years to increase immunization coverage rates. In addition, federal, provincial and territorial governments have worked together to establish a five-year set of objectives for the National Immunization Strategy that can capitalize on this new federal investment, leverage momentum and provide focus to their collective efforts.
The National Immunization Strategy Objectives 2016-2021 include:
- Setting national vaccination coverage goals and vaccine-preventable disease reduction targets
- Understanding barriers to vaccine uptake in under-immunized populations
- Ensuring timely and equitable access to vaccination services
- Developing evidence to inform effective program planning, with a view to increasing vaccination coverage
Goals for vaccination coverage and vaccine-preventable diseases reduction targets have been endorsed by provincial, territorial and federal governments via the Public Health Network Council. Progress towards reaching the national vaccination coverage goals and vaccine-preventable disease reduction targets will be reported every two years, using national coverage surveys and vaccine-preventable disease surveillance systems.
- Canadians now have access to an online and mobile app to keep track of their family's vaccinations with the CANImmunize mobile app. CANImmunize is a free, bilingual tool that is available nationwide.
- Methods used for national vaccination coverage surveys have been revised to measure vaccine hesitancy, barriers to vaccination, and to include First Nations (living off reserve), Inuit, and Metis children under two years of age.
- The mandate of the National Advisory Committee on Immunization includes scientific and economic evaluation, which will help to make vaccination program decision-making faster.
- The Canadian Institutes for Health Research in partnership with the Public Health Agency of Canada launched a funding opportunity for research to identify under- and non-immunized populations, and the socio-structural barriers associated with lower immunization access and uptake, in the Canadian population, with a particular focus on childhood immunization.
- The Canadian Immunization Research Network supports many research projects on vaccination issues. The Network will receive $10 million between 2017 and 2021. Of this, $2 million will be used for research on vaccination acceptance and uptake.
- A new Immunization Partnership Fund was launched to support provinces and territories and other partners in carrying out projects that improve vaccination access and uptake by identifying key barriers to vaccination and implementing or scaling up known best practices.
It is expected that these combined initiatives will contribute to improving vaccine access and uptake in Canada to reach our national vaccine coverage goals and disease reduction targets by 2025.
| Province/Territory | Web information |
|---|---|
| British Columbia | British Columbia ImmunizeBC |
| Alberta | Alberta Health Services Immunization |
| Saskatchewan | |
| Manitoba | |
| Ontario | |
| Quebec | Vaccination |
| New Brunswick |
|
| Nova Scotia | |
| Prince Edward Island | |
| Newfoundland | Newfoundland Labrador Health and Community Services Immunization |
| Yukon | Yukon Health and Social Services Immunization Information |
| Northwest Territories | NWT Health and Social Services Immunization/Vaccination |
| Nunavut | Nunavut Department of Health Immunization |
D1: National laboratory system
Joint external evaluation target: Real-time bio surveillance with a national laboratory system and effective modern point-of-care and laboratory-based diagnostics.
Level of capability in Canada
This report focuses on microbiology laboratories responsible for testing of biological samples. The laboratory system in Canada includes facilities at the national level, within each of the 13 provinces and territories, and at the local level. The National Microbiology Laboratory (NML) currently has four lab-based facilities.
At the provincial level, most jurisdictions have an officially designated provincial public health laboratory that often operates with close linkages to post-secondary institutions to support education and the ad